Heart Failure: An underappreciated complication of diabetes. A consensus report of the American Diabetes Association aims to improve diagnosis and hence slow progression of heart failure (HF) by increasing access to optimal management (Pop-Busui et al, 2022). It highlights benefits of multidisciplinary working, appropriate referral from primary care and shared care working. Although management options are similar to those in people without diabetes, the report provides a useful overview of HF, and summarises the evidence for drugs used to manage both the HF and hyperglycaemia.
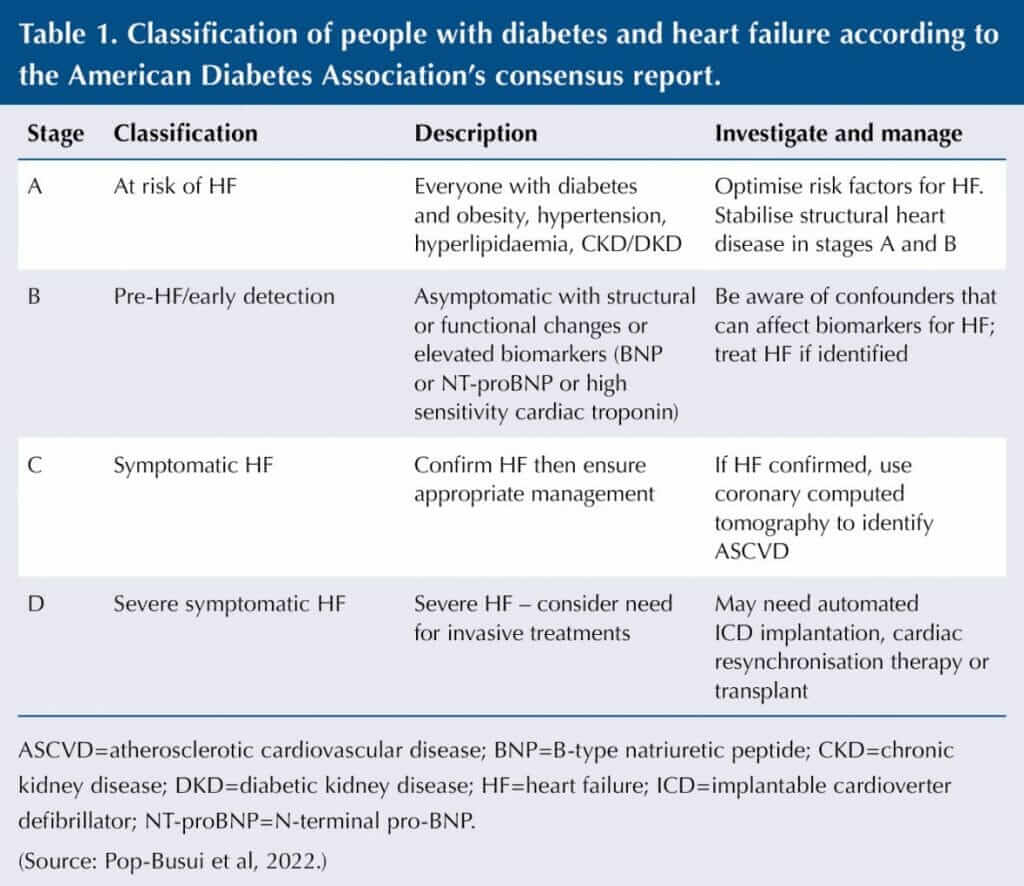
The consensus classifies people with diabetes and heart failure (see Table 1). Detailed history for signs and symptoms (e.g. exertional dyspnoea, fatigue and oedema) and clinical examination (weight, oedema, heart and lung exam) identify the need for bloods (including biomarkers), chest X-ray, ECG and transthoracic ECHO.
Annual HF biomarkers (BNP, NT-proBNP or high-sensitivity troponin) are recommended in those with diabetes, as HF is underdiagnosed. Elevated levels must prompt appropriate treatment and referral, while normal biomarkers have a strong negative predictive value so can reduce the need to pursue HF using other investigations.
Background
A 2–4-fold increased risk of HF has been demonstrated in large observational cohort studies in those with type 1 or 2 diabetes or pre-diabetes, and people with diabetes have a 3-fold increased risk of hospitalisation for heart failure and around 1.5-fold increase in mortality compared to those without. Duration of diabetes, glycaemic control, insulin resistance and female sex are strongly associated with incident HF, although it can also occur in those newly diagnosed or younger people. 10-year data from the UKPDS demonstrated HF incidence rates of 11.9/1000 patient years (Wamil et al, 2021). The relationship is bidirectional, with more than 60% of people with HF having insulin resistance, 20–30% having dysglycaemia, and a high incidence of new diabetes amongst people with HF, which is higher in Black, Hispanic, Native American and Asian groups.
Diabetic cardiomyopathy, defined as left ventricular systolic or diastolic dysfunction in the absence of other causes (hypertension or coronary artery disease) can occur. Both HF with reduced ejection fraction (HFrEF) and HF with preserved ejection fraction (HFpEF) are more common with diabetes or pre-diabetes. Glucose and lipid abnormalities can affect the myocardium, causing injury with detrimental effects on remodelling and function.
Management of HF in diabetes
Lifestyle
- The American Heart Association’s Life’s Essential 8 checklist – blood pressure, cholesterol, blood sugar, physical activity, eat well, lose weight, stop smoking/minimise alcohol and sleep well.
- Regular potassium monitoring, avoiding high potassium foods and drinks (including potassium-based salt substitutes), and drugs that can elevate potassium (e.g. NSAIDs).
- Consider individualised nutrition guidance, including DASH (Dietary Approaches to Stop Hypertension) or Mediterranean eating patterns.
- Individualised regular physical activity plan.
- Weight loss may reduce HF risk and improve other risk factors.
Control of modifiable risk factors for HF (such as poor glycaemic control, uncontrolled hypertension, hyperlipidaemia, obesity and microalbuminuria) is known to be suboptimal in people with diabetes, so primary care has an important preventive role at each clinic visit.
Pharmacologic treatment
Hypertension
- ACEi or ARB preferred, especially if albuminuria or coronary artery disease.
- Thiazide diuretic or ACEi more effective than calcium-channel blocker in preventing progression to symptomatic HF.
- Diabetes and diabetic kidney disease (DKD) but not symptomatic HF – finerenone may reduce progression of DKD and new HF.
Guideline-directed medical therapy (GDMT) for stages C and D
Similar to recommendations for those with HF without diabetes.
HFrEF
- RAAS blockade – combined sacubitril/valsartan (neprilysin inhibitor plus ARB, ARNI) or ACEi or ARB.
- Steroidal MRA – spironolactone or eplerenone.
- Beta-blocker – bisoprolol, metoprolol, carvedilol.
- SGLT2 inhibitors – currently dapagliflozin and empagliflozin have specific evidence in HFrEF.
- Ivabradine (to slow heart rate), hydralazine combined with isosorbide dinitrate (instead of ARNI, ACEi or ARB, especially in Black individuals or those with hyperkalaemia). Minimise dose of loop diuretics and optimise other therapies.
- Cardiac rehabilitation programmes – optimise referral and uptake as people with diabetes are less likely to access.
HFpEF (left-ventricular ejection fraction [LVEF] >40%)
- Previously, focus only on hypertension management with ACEi/ARB, loop diuretics for congestion and treatment of precipitating factors.
- LVEF <57% – spironolactone and sacubitril/valsartan now recommended.
- SGLT2 inhibitor – empagliflozin and dapagliflozin have demonstrated benefit in this group, and empagliflozin is licensed in UK.
Hyperlipidaemia
- Use statins based on age and risk factors.
Hyperglycaemia
- Individualised glycaemic targets recommended in those with diabetes and HF.
- SGLT2 inhibitors – exact mechanisms for benefit still being explored. Prioritise in those with stage B and use in all with symptomatic HF (stages C and D), if no contraindications.
- Consider GLP-1 RA (no direct HF risk reduction, but beneficial effects on weight, blood pressure and atherosclerotic cardiovascular disease), metformin or insulin, rather than sulfonylureas, in those with type 2 diabetes and HF requiring additional glycaemic control. Stop metformin if acute conditions associated with lactic acidosis.
- DPP-4 inhibitor or thiazolidinediones are not recommended in those with type 2 diabetes and stages B, C or D heart failure.
Metabolic surgery
- Improves risk factors and cardiovascular events.
Hospitalisation
- Highlights worse prognosis. 90-day readmission rate and 1-year mortality of 30%, so treat these people as priority.
- Ensure in-hospital medication changes are continued and HF drug doses are up-titrated as recommended and tolerated. Shared care with community HF teams helpful.
Consider referral
- Stage A – for risk factor management if needed.
- Stage B – for global risk assessment, investigation (e.g. ECHO), exploration of causes and initiation of therapies.
- Stages C and D – shared care for titration of therapies can be useful.
The consensus concludes with a lengthy list of knowledge gaps and future research questions.
A recent post hoc analysis of the ACCORD Lipid study demonstrated that treatment with simvastatin plus fenofibrate, compared with simvastatin and placebo, significantly reduced the risk of hospitalisation for heart failure (HHF) and cardiovascular death by 18% overall (Ferreira et al, 2022). Further analysis demonstrated a 36% reduction in those in the standard glycaemic control group (HbA1c 7–7.9% [53–63 mmol/mol]), with no benefit in those randomised to intensive control (<6% [<42 mmol/mol]). This reduction in risk is comparable to that in those treated with SGLT2 inhibitors. However, Morieri (2022), writing in an accompanying editorial, warns caution is required, as the mechanism for these differential effects is unclear and effects in people already on the preferred SGLT2 inhibitor therapy may differ. Hence, the study should be seen as hypothesis-generating only. Fenofibrate is an agonist of peroxisome proliferator-activated receptor-alpha (PPAR-alpha), known to regulate vascular inflammation, cell differentiation and cardiac energy metabolism.
Click here for the full consensus report.
Click here for the Morieri editorial





The mortality benefits of smoking cessation may be greater and accrue more rapidly than previously understood.
2 Apr 2024