What and why
Incretin mimetics are licensed in the UK for the management of type 2 diabetes and obesity. Commonly used medications include semaglutide (Ozempic, Wegovy and Rybelsus), tirzepatide (Mounjaro), liraglutide (Saxenda) and dulaglutide (Trulicity).
Other, potentially more potent, incretin-based medications are also emerging, including oral GLP-1 receptor agonists; dual agonists of GLP-1/GIP, GLP-1/glucagon or GLP-1/amylin; and triple agonists of GLP-1, GIP and glucagon.
Although Sattar et al (2025) and others argue that muscle loss observed with these therapies is broadly proportional to overall weight loss and is not excessive compared with other weight loss methods,1 concerns have been raised that GLP-1 receptor agonists may lead to disproportionate muscle loss.
Preserving muscle mass remains clinically important, and both NICE and the World Health Organization recommend wraparound care to support nutrition, physical activity and long-term health behaviours.
Healthcare professionals have a responsibility to discuss the risk of muscle loss as a potential side-effect of significant weight loss, including when using incretin mimetic therapies, and to provide guidance on mitigation strategies.
Muscle loss and sarcopenia in type 2 diabetes
Definitions
Muscle loss is a broad clinical term describing a reduction in skeletal muscle mass resulting from various causes. Sarcopenia is a progressive condition affecting skeletal muscle, characterised by declining muscle mass, strength and functional capacity.
Muscle loss is clinically classified as sarcopenia only when the reduction in muscle quantity is accompanied by a measurable decline in muscle strength and/or physical performance.2
Lean mass is frequently used as a proxy for muscle mass in research studies, although it is not synonymous with skeletal muscle, as it also includes water, organs and other non-fat tissues. Note that reductions in lean mass during weight loss do not necessarily equate to clinically meaningful muscle loss unless accompanied by declines in strength or physical performance. Increasingly, muscle quality (including fat infiltration and metabolic function) is recognised as an important outcome, and may improve even where absolute muscle mass decreases.
Causes
Muscle loss is multifactorial and may result from:
● Increasing age: Age-related muscle loss involves neuromuscular decline, reduced motor unit recruitment and increased anabolic resistance. Muscle mass typically declines by approximately 0.3–0.5% per year after the age of 30, increasing to up to 1% per year after the age of 60.3,4
● Metabolic disease: In type 2 diabetes and obesity, insulin resistance impairs muscle protein synthesis, while chronic inflammation, fat infiltration and mitochondrial dysfunction further compromise muscle health.
- People living with type 2 diabetes have a two- to three-fold higher risk of developing sarcopenia than those without diabetes and are at increased risk of accelerated ageing and frailty.5
● Significant weight loss (regardless of the method used): Reductions in muscle mass are driven by energy and protein deficits, increased muscle protein breakdown and reduced mechanical loading.
- Dietary weight loss interventions typically result in approximately 20–30% of total weight loss being derived from lean mass.
- Evidence from studies of GLP-1 and GIP/ GLP-1 receptor agonists suggests that reductions in lean mass are broadly proportional to overall weight loss; however, estimates vary depending on the method of assessment and study population.1,6
● Reduced physical activity or increased sedentary time (particularly among older adults experiencing falls, hospital inpatient stays or neurodegenerative conditions).
Mechanisms behind muscle loss
Muscle loss due to ageing, diabetes (type 1 or type 2), obesity or significant weight loss shares a number of key mechanisms, including:
- Hormonal changes (reduced anabolic hormones, including testosterone, growth hormone and IGF-1, which normally support muscle protein synthesis and repair).
- Loss of muscle fibres.
- Oxidative stress.
- Chronic inflammation.
The rate of muscle mass decline varies widely depending on age, disease duration, lifestyle factors and assessment method.
Early and effective interventions can play a key role in preventing, delaying, and in some cases partially reversing sarcopenia, particularly in individuals at increased metabolic risk.
Who is at risk of muscle loss?
Individuals at increased risk of clinically significant muscle loss include the following:7
- Older adults, particularly those aged over 65 years.
- Frailty, recent hospitalisation or prolonged inactivity.
- Low baseline muscle strength or sarcopenic obesity.
- Rapid weight loss, very low energy intake or persistent gastrointestinal side-effects.
- Comorbidities that limit physical activity (e.g. osteoarthritis, neuropathy, cardiovascular disease).
- Chronic kidney disease.
These risk factors are particularly relevant when initiating weight-loss interventions, including GLP-1 and GIP/GLP-1 receptor agonist therapies, where reduced appetite and rapid weight loss may further increase vulnerability to muscle loss. These groups may require individualised nutritional planning.
The impact of sarcopenia2
Increased mortality
People with sarcopenia have a 2-times higher risk of death compared to those without the condition.8
Functional decline and disability
Sarcopenia strongly predicts loss of physical function and reduced ability to perform activities of daily living. Declining strength and mobility increase dependency over time.
Higher risk of falls
Multiple studies demonstrate an association between sarcopenia and increased fall risk in older adults. Falls are a major contributor to injury, hospital admission and loss of independence.
Hospital admissions and healthcare utilisation
Sarcopenia is associated with higher rates of hospital admission and potentially increased lengths of stay.
Fragility fractures (less consistent evidence but clinically important)
Some evidence suggests an association between sarcopenia and fracture risk, likely mediated through muscle weakness and falls, although prospective cohort data is less consistent.7
Reduced quality of life
Sarcopenia negatively impacts health-related quality of life, affecting both physical and mental wellbeing due to reduced independence, mobility and strength.9 While this is well documented in the general population, the presence of type 2 diabetes significantly exacerbates these risks.
Benefits of maintaining muscle mass
● Supports physical function, mobility and independence across the lifespan.
● Contributes to metabolic health, including improved glucose regulation and reduced cardiometabolic risk.
● Helps preserve bone density and reduces the risk of falls and fractures.
● Plays a key role in maintaining strength, balance and postural stability.
● Supports mental health, cognitive function and overall quality of life.
● Helps preserve resting energy expenditure during weight loss.
● Reduces the risk of weight regain following weight loss.6
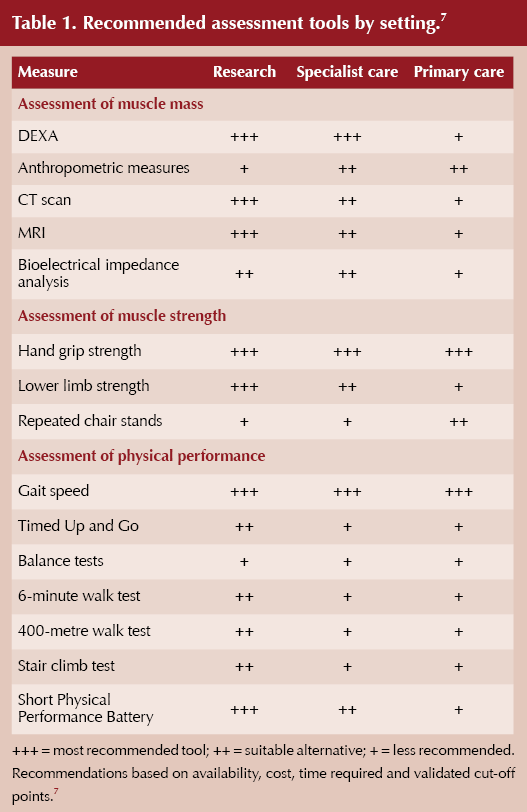
Assessment of muscle mass
Assessment should consider overall health, frailty risk, functional status, and the individual’s capacity to engage with advice in primary care.
Simple screening tools such as the SARC-F questionnaire can be used in primary care to identify individuals at risk of sarcopenia. This tool assesses strength, assistance with walking, rising from a chair, climbing stairs and risk of falls, and provides a quick, low-cost method for case-finding.10
Some assessments that can be carried out in primary care lack the sensitivity and specificity of image-based assessments such as DEXA scans; however, they might be useful, quick, low-cost and practical options to enable early identification of individuals at risk of muscle loss without the need for specialist assessment (Table 1).
Care planning: A practical guide
It is important to highlight the potential risk of muscle loss with incretin-based medications and ways to minimise it. All individuals on these medications benefit from a supervised, structured exercise programme, as these programmes offer the strongest evidence for preserving muscle and supporting weight maintenance after therapy.11
Strength training (2–3 times per week)
● Aim for at least 10 minutes per session, 1–2 times a week initially.
● Stagger resistance training across the week (improves glucose regulation in type 2 diabetes by capitalising on post-exercise insulin sensitivity, which remains elevated for 48 hours).
● Focus on major muscle groups: legs, back, chest and core.
● Examples include bodyweight exercises, resistance bands, light weights, Pilates, gym-based or seated exercises.
● Encourage slow, controlled movements and gradual progression (systematically increasing frequency, intensity, time and type to ensure continuous physiological adaptation).
● Consider referral to diabetes-specific weight management or exercise programmes, where available.
Daily movement
● Encourage light walking, stretching or regular movement breaks.12,13
● Aim towards 150 minutes of moderate-intensity activity per week, where possible.
● Start low and progress slowly; even an additional 500 steps per day can improve cardiometabolic health.
Hydration
● Encourage regular fluid intake, aiming for approximately 2–3 litres per day, unless clinically contraindicated.
Protein intake
● Aim for 1.0–1.2 g per kg body weight per day as general guidance.14,15
- Consider higher intakes (≥1.2 g/kg/day) in older adults, frailty, illness or with resistance training.
- Higher protein intakes should be considered with caution in individuals with impaired renal function, and may require individualised advice or dietetic referral.
● Encourage protein at each meal/snack (e.g. eggs, Greek yoghurt, tofu, chicken, fish, beans, cottage cheese).
● Combine with fibre-rich foods and healthy fats to support overall nutrition.
● Adjust advice based on appetite, weight loss rate and tolerance.
● Promote balanced meals, including appropriate portions of complex carbohydrates (e.g. oats, brown rice, quinoa, wholewheat pasta, lentils, potatoes, wholemeal bread), vegetables, salad and fruit.
Author’s recommendation
● Personalise care plans based on the individual’s goals, abilities, priorities and preferences. Use of shared decision-making approaches is essential to support sustainable weight loss and long-term weight maintenance.
Example individual patient plan
Use these prompts to support SMART goal-setting. Ask the person to choose specific goals that are measurable, achievable and realistic for them, then decide on a time frame and ensure timely follow-up. It is crucial to remember that people don’t have to set a goal for each point; one small goal may be enough initially.
Strength plan
e.g. Two days per week: resistance-band routine at home; attend a yoga class.
Daily movement goal
e.g. 10–15-minute walk after meals, or an additional 500 steps/day; get up during TV advert breaks for a walk/stretch.
Protein focus
e.g. Include a protein source at every meal; add a Greek yoghurt snack.
General nutrition
Aim for a variety of vegetables and salad (target five portions/day where possible), 1–2 portions of fruit and modest portions of complex carbohydrates. Discuss affordable options, such as tinned and frozen produce, where relevant.
Sleep
Aim for 6–8 hours of good-quality sleep (e.g. going to bed 30 minutes earlier).
Other goals
Hydration reminders, identifying barriers, coping with lapses, planning for eating out, meal ideas, exploring alternative ways to be physically active in those with contraindication to regular movement, and signposting to Diabetes UK or other trusted resources.
Advice to minimise gastrointestinal side-effects
Gastrointestinal side-effects with incretin‑mimetic drugs are most common during initiation and dose escalation, so minimising them involves slow dose titration with patient education.
- Dietary adjustments include eating smaller portions and low‑fat, easy‑to‑digest meals; eating slowly; increasing hydration; and adjusting portion size to tolerance.
- If symptoms persist, dose slowing or temporary symptomatic therapies may be considered.16
Acknowledgement
The author thanks Dr Matthew McCarthy at Leicester Diabetes Centre for his feedback on this article.





Jane Diggle reflects on the growing role of incretin-based therapies across cardio–renal–metabolic care, and highlights resources to support their use.
12 May 2026