Around 60 000 new type 2 diabetes cases missed in 2020
Matthew Carr (University of Manchester) examined the indirect effects of the COVID-19 pandemic on the diagnosis and monitoring of type 2 diabetes in 2020, using anonymised primary care data from the Clinical Practice Research Datalink (CPRD). Compared with historical data from the previous 10 years, there was a 70% drop in the rate of new type 2 diabetes diagnoses in April 2020 and, while the number of diagnoses gradually rose to average levels by the end of the year, they were greatly reduced throughout the majority of the year, and this shortfall has yet to be addressed.
Extrapolating these data to the whole of the UK, an estimated 60 000 new type 2 diabetes diagnoses have been missed, and this may even have been an underestimate if changes in diet and physical activity during the various lockdowns have led to an above-average incidence of the condition.
The drop in diagnoses was largest in England compared with the other UK nations; however, the same pattern was observed across the UK. New metformin prescriptions followed the same pattern, suggesting that the issue is genuinely one of missed diagnoses rather than a lack of coding.
Among people with pre-existing type 2 diabetes (approximately 600 000 captured within the CPRD), similar reductions emerged when analysing rates of the key care processes and treatment escalation (new prescriptions of glucose-lowering and antihypertensive drugs). In contrast, repeat prescription rates did not fall.
Given the consequences of delayed diagnosis on diabetes-related complications, there is an onus to resume health checks and diabetes screening as quickly as possible. A new Quality and Outcomes Framework indicator, The percentage of patients with non-diabetic hyperglycaemia who have had an HbA1c or fasting blood glucose performed in the preceding 12 months, should help motivate practices to undertake this task.
The findings have since been published in The Lancet Diabetes & Endocrinology (click here to access).
Counterbalance study: Cardiovascular effects of weight loss in type 2 diabetes
New results from the DUK-funded Counterbalance study reveal that putting type 2 diabetes into remission through weight loss can have the additional benefit of reducing the risk of cardiovascular diseases such as a heart attack or stroke (Poster P25).
Thirty participants underwent an 8-week very-low-calorie diet followed by a stepwise return to an isocaloric diet and a 6-month weight maintenance phase. At 6 months, mean weight fell from 98 kg to 85 kg, and 13 participants achieved remission (HbA1c <48 mmol/mol and fasting plasma glucose <7.0 mmol/L after withdrawal of glucose-lowering drugs at the start of the trial).
Ten-year cardiovascular risk (QRISK3 model) decreased significantly after weight loss both in those who achieved remission and in those who did not, but non‐responders remained at higher risk (15.0% vs 5.8%). At 6 months, body weight and subcutaneous and visceral fat volumes decreased in both groups, and plasma biomarkers of cardiovascular risk improved after weight loss, irrespective of remission status.
The authors concluded that weight loss markedly decreases cardiometabolic risk, particularly when remission of diabetes is achieved. Normalisation of 10-year cardiovascular risk and heart age is possible after substantial dietary weight loss, and particularly if remission of diabetes is achieved.
Conducting difficult diabetes consultations
Marilyn Ritholz (Joslin Diabetes Center, Boston, MA, USA) outlined the latest research on the interactions between healthcare professionals and people with diabetes, paying particular attention to the difficult task of explaining the need to prevent microvascular complications such as retinopathy and renal disease. The key implications for practice were as follows:
- Conduct individualised and compassionate conversations that include thorough and complete information and specific self-care guidance.
- Avoid messages of fear, blame and judgement, and do not use scare tactics.
- Find the right balance of being open and honest but also providing hope that complications can be avoided with successful self-care.
- Promote active self-care as soon as possible after diagnosis, but taking into account the individual’s (or their family’s in the case of children and young people) capacity to handle simple versus complex information.
- Inquire about and address the individual’s social and emotional needs.
Age, gender and ethnicity barriers to FreeStyle Libre access
Analysis of the FreeStyle Libre audit conducted by the Association of British Clinical Diabetologists suggests that older people, men and people of non-white ethnicity are less likely to be using the flash glucose monitoring device than their counterparts (Poster P211).
Compared with the type 1 diabetes population in the National Diabetes Audit (NDA), in the Libre audit, men were significantly under-represented (48.1% vs 56.5% in the NDA), as were black people (0.2% vs 2.7%) and Asian people (2.0% vs 4.3%). People with a shorter diabetes duration and those over 60 years of age were also under-represented.
As uptake of the Libre audit is not uniform across the country, it is possible that these findings merely reflect regional variation in type 1 diabetes populations; however, it seems likely that these groups face barriers to taking up flash monitoring. Given the potential impact on glycaemia, hospitalisation and diabetes distress that the technology has, it is important to overcome these barriers.
Type 1 diabetes and disordered eating (T1DE)
This session introduced the concept of type 1 diabetes and disordered eating (T1DE), including anorexia, bulimia, binge eating disorder and insulin omission to reduce weight. T1DE is not yet recognised as either a medical or a psychological disorder. However, while the term “diabulimia” is more widely used, this is insufficient to describe the wide array of phenotypes and clinical presentations of type 1 diabetes-associated disordered eating.
Outlining the work of the NHS England-funded ComPASSION (Combined Pathway for Assessment and Support for the Syndrome of Insulin OmissioN) project, Carla Figueiredo and Nicola Stacey (Dorset Healthcare NHS Trust and Royal Bournemouth Hospital) provided a working definition to diagnose T1DE (Box 1). They went on to describe the work of the ComPASSION project in creating multispecialist teams using joined-up working between diabetes and eating disorder multidisciplinary teams.

Marietta Stadler (King’s College London) spoke about the STEADY (Safe management of people with Type 1 diabetes and EAting Disorder studY) programme to develop novel interventions for people with T1DE. To date there is no evidence-based intervention to improve glycaemic control in this population.
Through a collaboration between people with type 1 diabetes, healthcare professionals, psychologists and dietitians, the STEADY team has developed a toolkit that draws on cognitive behavioural therapy and diabetes education, and which can be tailored to the needs of individual patients (Poster P238). As a next step, the toolkit will be tested in a feasibility randomised control trial to compare it with standard care and to refine it.
Metformin in pregnancy: Pros and cons
Fidelma Dunne (National University of Ireland Galway) outlined the evidence for and against using metformin in pregnancy, in women with either pre-existing type 2 diabetes or gestational diabetes. Although its use in pregnancy is an off-label indication, metformin has a wealth of safety data from its use over the last 60 years, it is simple to use and generally well tolerated, and it has a low cost.
Large international registry studies have found no evidence of any increased risk of congenital abnormalities with first-trimester exposure to metformin therapy. Randomised controlled trials have shown short-term pregnancy benefits compared with placebo, including lower rates of excessive maternal weight gain and improvements in glycaemic control, as well as lower rates of macrosomia and neonatal hypoglycaemia.
In a direct comparison with insulin in women with gestational diabetes, the metformin group had better maternal postprandial glucose and less maternal weight gain, with less neonatal hypoglycaemia. Maternal preference was also greater for metformin, and medication concordance was higher.
However, while metformin reduces the likelihood of large-for-gestational-age offspring, it also increases the rate of small-for-gestational-age children. Longer-term follow-up at 2–10 years of age also showed that children exposed to metformin in the womb had higher mean BMI and other measures of adiposity. Compared with insulin, there was a higher risk of preterm birth with metformin.
When asked for her opinion, Professor Dunne suggested she would avoid metformin in cases of fetal abnormalities, small fetal size (<10th centile), twins, or abnormal renal or liver function in the mother. She would also suspend treatment if fetal growth slowed and fell below the 10th centile. She also suggested taking a detailed history of the mother and avoiding metformin if there was any suspicion of type 1 or monogenic diabetes.
Weight management issues
Rachel Batterham (University College London) outlined the barriers that people with type 2 diabetes and excess weight face in accessing treatment. These include low prioritisation and funding for weight management services, a widespread lack of understanding of the physiology of obesity, and the stigma that results. She highlighted a number of ways healthcare professionals can help.
Signposting to patient support groups is a highly effective way to help overcome barriers to treatment. Such groups include Obesity UK, the European Coalition for People Living with Obesity, the Obesity Action Coalition and the Obesity Empowerment Network UK.
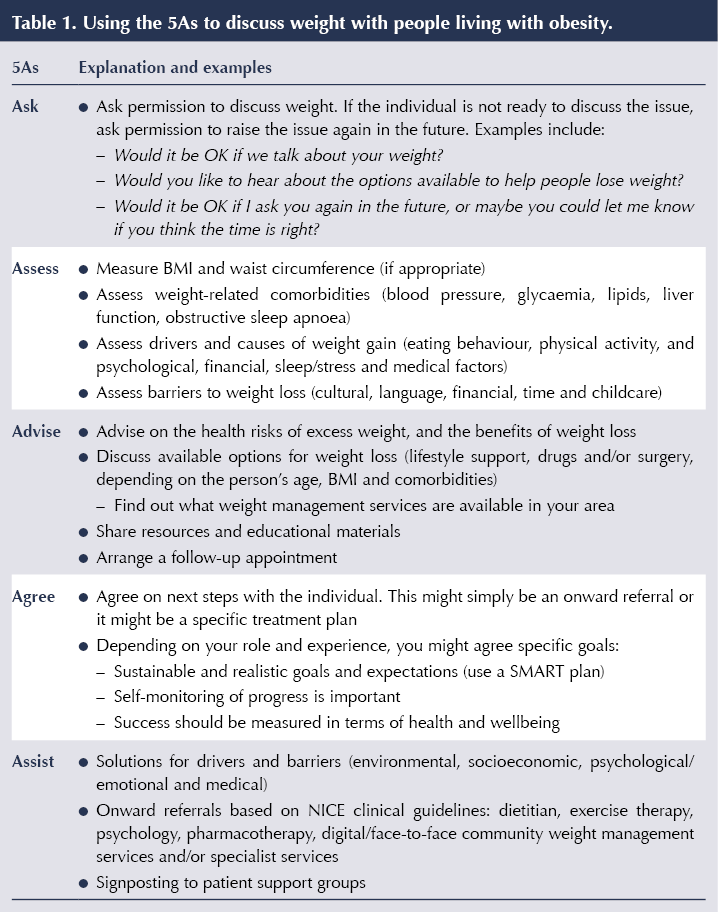
Professor Batterham also recommended that healthcare professionals use the 5As strategy (Ask, Assess, Advise, Agree, Assist) to discuss weight with their patients (Table 1).





Developments that will impact your practice.
26 Jun 2025