Although mortality rates from cardiovascular disease (CVD) are decreasing, many individuals live with multiple risk factors, including hypercholesterolaemia that is not well controlled (Ferrières et al, 2016). Achieving recommended levels of LDL cholesterol using currently available oral lipid-lowering therapies is even more challenging in people with obesity or diabetes (Robinson et al, 2015). Increasingly, novel strategies in the form of injectable medicines are being developed and approved to reduce the risk of cardiovascular events in people at high risk of, or with established, CVD. Despite being highly effective, however, these agents remain underused (Ray et al, 2023; Scheen et al, 2024).
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors are a class of lipid-lowering therapy which can be offered for primary prevention of CVD among people with familial hypercholesterolaemia, or for secondary prevention in people with established atherosclerotic CVD who need further lowering of their LDL cholesterol or who are intolerant to statin therapy (Landmesser et al, 2017; Sabatine et al, 2017; NICE, 2023). Randomised controlled trials have demonstrated the effectiveness of PCSK9 inhibitors in lowering cholesterol and improving survival rates in secondary prevention (Lipinski et al, 2016; Baylis et al, 2017; Ridker et al, 2017; Sabatine et al, 2017).
For those with CVD and diabetes, injectable glucose-lowering medicines, such as liraglutide, semaglutide and tirzepatide, have been shown to reduce risk of cardiovascular events in addition to their effects on HbA1c (Marso et al, 2016a; Marso et al, 2016b; Nicholls et al, 2025). Semaglutide has also demonstrated benefits in people with obesity and long-term benefits to the kidneys (Colhoun et al, 2024; Perkovic et al, 2024).
The need for education
Previous studies have explored patient barriers to the use of injectable therapies for cardiovascular conditions, including reluctance to use injectable therapies and lack of education about their indications for use and clinical benefits (Lee et al, 2022). For those with diabetes, barriers to insulin use still exist, and a systematic review highlighted issues relating to three areas: patients, healthcare professionals and system factors (Ng et al, 2015).
Reluctance to offer injectable therapies has also been observed among clinicians, who are sometimes hesitant to intensify therapy using injectable agents such as insulin and PCSK9 inhibitors (Brod et al, 2014; Ng et al, 2015). In an anonymous survey involving 192 healthcare professionals, commonly raised issues were resource problems, lack of knowledge among colleagues, paperwork and lack of patient knowledge (Khatib et al, 2022). Key barriers that respondents felt made patients decline these treatments were fear of injections, lack of awareness or education, and administration issues; potential reasons for discontinuation included side-effects, perceived lack of benefit and local reactions. The main topics around injectables requiring further support included managing non-adherence, troubleshooting with patients and educating colleagues about injectable therapies.
Assessing individual needs and identifying and reducing any physical or psychological barriers to injectable medication may help facilitate earlier introduction of therapy into the CVD treatment plan, thereby assisting patients and their families to achieve optimal control of CVD risk factors and increasing adherence to these new therapies (McKee et al, 2014; Klindtworth et al, 2015; Saborowski et al, 2018). Healthcare professionals, particularly nurses and pharmacists, have a vital role not only in the management of injectable therapies in CVD, but also in informing, educating and coordinating patient care in such settings (García-Pérez et al, 2013; Brod et al, 2014; Ng et al, 2015). As their roles are integral to the healthcare pathway of people with CVD, it is critical that healthcare professionals are also well informed and confident in the management of injectable therapies.
Based on the previous findings around the perception of injectables with cardiovascular benefits (Lambrinou et al, 2020), the present authors, in conjunction with the European Society of Cardiology (ESC), have developed educational materials, in different multimedia formats, that encompass specific educational programmes focused on patients’, caregivers’ and healthcare professionals’ needs.
Development of educational materials
Two educational leaflets and two videos, targeting patients/carers and healthcare professionals separately, were developed, optimised, translated into French and Italian, and published on a dedicated ESC website, available at: https://bit.ly/4sBIpPJ (Figure 1).

(A) Homepage (includes videos and education leaflets [English, French, Italian], articles and slides).
(B) Electronic educational leaflets for healthcare professionals and patients.
Initially, multiple initial leaflet drafts were created with different layouts and limited visuals. They were more academic, focusing on the evidence. Following a number of key stakeholder meetings and feedback from a variety of professionals, several priorities were identified according to the target audience. For healthcare professionals, more support on identifying eligible patients, prescribing, monitoring, promoting adherence, troubleshooting and accessing resources for injectables with cardiovascular benefits was needed.
After discussions in focus groups and feedback through an online survey involving patients, carers and members of the public, the leaflets were optimised to include only the core information on injectables, with more engaging visuals and colours. The final versions were focused on practical advice on the management of injectables for both target audiences: patients/carers and healthcare professionals.
With regard to the educational videos, one was created for patients/carers and one for healthcare professionals. Three volunteer patients on injectables with CVD benefits (PCSK9 inhibitors and GLP-1 receptor agonists) were identified, with diversity in age, gender and ethnicity. Their consent was obtained during the day of filming, which was facilitated by the local videography team, who also edited the videos. Among the discussion topics were the indications for injectable therapies, information sources (healthcare professionals, internet, peers, etc.), the ease of use, dosing frequency, side-effects, monitoring and their impact on cholesterol, especially LDL, reduction. The final patient video lasts around 6 minutes.
For the healthcare professional video, five specialist clinicians from Leeds Teaching Hospitals NHS Trust were interviewed on lipid optimisation. Discussion included an overview of injectable therapies, barriers and myths, and suggested actions and strategies to improve uptake. The filming lasted for about 2 hours and the final video was edited down to around 7 minutes, following multiple rounds of montage.
Assessment of acceptability of materials
All materials were examined by volunteer reviewers for their user-friendliness, accessibility and ease of understanding the information provided. This was particularly important for the patient materials, which went through multiple rounds of scrutiny (using a number of online accessibility tools and expert reviews) for their accessibility by non-native English speakers across Europe. Overall, feedback was positive, with areas for improvement also identified.
Patient leaflet and video
Two rounds of patient and public involvement and engagement activities with members of a variety of local groups with lived experience of CVD, with or without experience of injectables, were held. A total of 27 people provided feedback for the leaflet and 25 for the video, with a mostly positive view of the materials (Figure 2). Of those, 74% had limited or no prior knowledge of these injectable therapies.
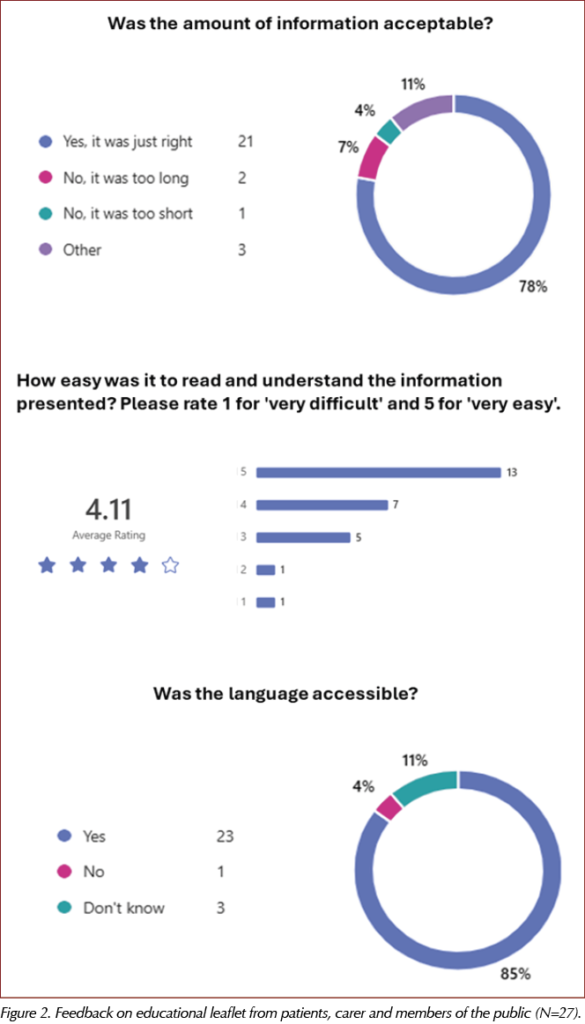
Prior to being exposed to the educational materials and providing feedback, positive attitudes were observed in those with healthcare backgrounds and those with previous experience of using injectables (26%). Thematic and sentiment analysis of the respondents’ first impression of injectables revealed that positive sentiments, combining practical benefits (i.e. convenience, alternative to oral medication) and positive experiences, outweighed safety concerns and fears (37% vs 16%). The review of educational materials did not aim to change the reviewers’ opinion about injectables in any way; nevertheless, the percentage of reviewers with a positive view towards them increased from 48% to 74%, demonstrating that the materials further strengthened their positive attitude.
Among the priorities identified and suggestions proposed by patients/carers were the following:
- 50% for content clarity and organisation (additional explanations of technical terms and structural suggestions).
- 42% for language and accessibility.
- 33% for visual and layout improvements (formatting and image-specific suggestions).
- 25% for more detailed medical and safety information.
Regarding content clarity, information on the proper use of injectables (40%), their safety profiles (30%), and their appropriate storage conditions and means of transportation (20%) was requested. Around 10% of participants were concerned about credible, independent safety data and the long-term use of injectables. As a result, they asked for trustworthy websites and reliable electronic resources to be included within the educational materials.
Healthcare professional leaflet and video
Similarly, for the materials targeting healthcare professionals, 24 clinicians commented on the educational leaflet and 10 on the video. Out of those reviewing the leaflet and the video, respectively, 46% and 40% were experienced in prescribing and counselling patients on injectables. The rest did not have specialist knowledge or use or manage these medications routinely.
Apart from some minor corrections and a mostly practical focus on content, the predominant suggestion of clinicians reviewing the materials for user-friendliness and acceptability was around the inclusion of particular guidelines; however, these were solely UK-based (i.e. NICE guidelines and technology appraisals). Due to the wider European audience that would ultimately be exposed to these materials, we have had to limit directing readers to CVD guidelines at the European level; such guidelines are traditionally and solely provided by the ESC anyway.
The second most common recommendation from clinicians was to expand on novel injectables (e.g. inclisiran, semaglutide, tirzepatide) that were not part of the scope of this project at the time of design. This feedback was considered very seriously and has since led to the design of a follow-up, UK-based injectables project with a component to develop additional educational materials that are more specific to those newer injectables.
Future directions
The need for educational resources for patients, caregivers and healthcare professionals to inform about the latest developments in this area continues to grow with the ever-expanding number of injectables in the market. In addition to inclisiran, tirzepatide and semaglutide, even more injectables are in the pipeline and will soon appear in the market. For example, survodutide (BI 456906; Boehringer Ingelheim), a dual glucagon/GLP-1 receptor agonist, is currently in Phase 3 clinical trials for the treatment of adults with non-cirrhotic metabolic dysfunction-associated steatohepatitis and moderate or advanced fibrosis (Sanyal et al, 2024; ClinicalTrials.gov, 2026). Additionally, most medications being developed to lower lipoprotein (a), another risk factor for CVD, are in injectable form. It is expected that injectable therapies will have a significant role to play in the cardio–renal–metabolic (CaReMe) space in the years to come.
Recently, reflecting on the appetite for additional educational resources as well as the great opportunity for staff development and upskilling as a result of involvement in the development of these materials, it was decided that a follow-up project would be designed. We have since concentrated our team’s efforts towards understanding the similarities and differences in barriers and facilitators between different injectables currently available in the UK. We have included the most novel injectables available in the UK, with the older classes still within our scope. Ultimately, the aim is to expand on the materials we have developed. While these are still up to date and very informative about the use of injectables generally and these classes specifically, differences in frequency of administration, characteristics and mechanisms of action between the older and newer classes of injectables may require further information to support their use and management.





Greater efficacy at the cost of more adverse events in this developmental oral GLP-1 receptor agonist.
2 Apr 2026