Quick summary of management updates
In general
● Reinforce advice about diet and healthy living at all stages.
● Assess obesity, cardiovascular status (including heart failure) and future risk, renal status and frailty risk.
● Discuss benefits and risks of each medicine and their impacts on HbA1c, weight, CVD and renal protection. Be aware of retinal risks if sudden decrease in HbA1c.
- Consider contraindications/factors which might influence medication choice.
- If there is more than one equally suitable medicine in the same class, choose the least expensive.
● If response to medicines suggests type 2 diabetes may not be the correct diagnosis, see the Initial diagnosis section of the NICE NG17 guideline on managing type 1 diabetes.
● Give clear sick day rules in each person’s individualised treatment plan. Follow MHRA safety advice on ketone monitoring with SGLT2 inhibitors.
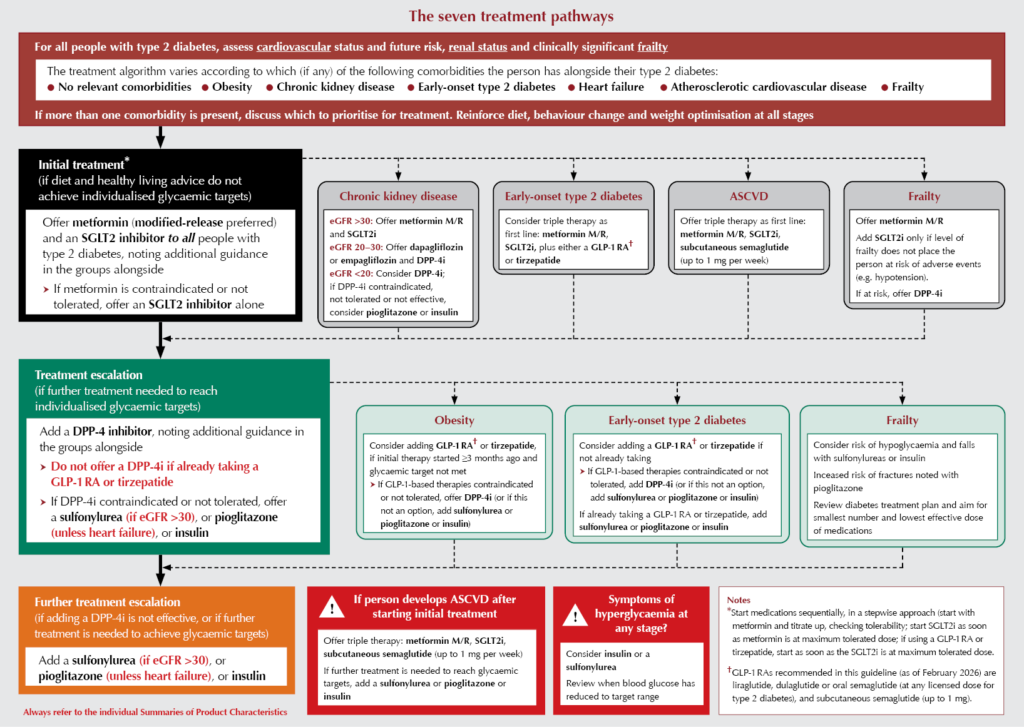
Seven-track algorithm (see algorithm below)
The treatment algorithm varies according to which (if any) of the following comorbidities the person has alongside their type 2 diabetes:
- No relevant comorbidities.
- Obesity.
- Chronic kidney disease.
- Early-onset type 2 diabetes.
- Heart failure.
- Atherosclerotic cardiovascular disease.
- Frailty.
If more than one comorbidity is present, discuss which to prioritise for treatment with the person.
First-line therapy
● Offer metformin modified-release (M/R) and SGLT2 inhibitor dual therapy first-line, even if no other comorbidity is present, unless contraindicated (caution with SGLT2is in those with frailty).
● Initiate metformin M/R in preference to standard-release metformin, unless the person has swallowing difficulties and needs to crush tablets or use liquid preparations.
- If eGFR is <45 mL/min/1.73 m2, reduce metformin dose to 1 g per day. Do not initiate if eGFR is <30.
- Can continue standard-release metformin if effective, but change if not tolerated or according to person’s preference.
● Titrate metformin M/R to check tolerability, then add SGLT2i.
● Use SGLT2i monotherapy if intolerant of metformin M/R.
SGLT2 inhibitors:
● Address inequalities in SGLT2 inhibitor access and uptake by:
- Monitoring who is using SGLT2 inhibitors.
- Identifying groups who are eligible but who have a lower uptake (evidence suggests these include women and older people, certain minority ethnic backgrounds and socioeconomically deprived groups).
- Making plans to engage with these groups to encourage them to use SGLT2 inhibitors.
● Before initiating SGLT2 inhibitors, check for previous diabetic ketoacidosis (DKA), risk of dehydration or volume depletion, or if following a very-low-carbohydrate or ketogenic diet. Address modifiable risks for DKA before initiating.
● Do not initiate while person is unwell with intercurrent illness.
Earlier use of GLP-1 RAs/tirzepatide
● Earlier use of a GLP-1 RA or tirzepatide, with fewer restrictions, is recommended:
● Offer subcutaneous semaglutide (up to 1 mg only) as first-line triple therapy if ASCVD.
● Consider a GLP-1 RA or tirzepatide as first-line triple or first additional therapy for those with early-onset type 2 diabetes.
● Consider a GLP-1 RA or tirzepatide as the first additional therapy for people with obesity who need further glucose-lowering treatment.
Triple therapy
● Offer triple first-line therapy (metformin, SGLT2i and subcutaneous semaglutide) for people with ASCVD.
- Titrate metformin, add SGLT2i, then add subcutaneous semaglutide and titrate up to 1 mg.
- Initiate drugs sequentially. Check tolerability before adding next medication or titrating.
● Consider triple first-line therapy (metformin, SGLT2 inhibitor and a GLP-1 RA/tirzepatide) for people with early-onset type 2 diabetes (onset at <40 years).
Additional therapies
● Offer a DPP-4 inhibitor if needed for additional glucose lowering (but prioritise semaglutide, GLP-1 RA or tirzepatide where these are recommended).
- Do not prescribe GLP-1 RAs or tirzepatide together with a DPP-4 inhibitor.
● If a DPP-4 inhibitor is contraindicated, not tolerated or not effective, pioglitazone (unless heart failure is present), a sulfonylurea (if eGFR is >30 mL/min/1.73 m2) or insulin can be added, depending on circumstances.
Continuing/stopping medications
● When people reach their glycaemic or weight targets, consider continuing medications which have contributed.
● Continue SGLT2is for cardiovascular and renal benefits even if not helping reach glycaemic targets.
● Stop GLP-1 RAs or tirzepatide if:
- Person becomes underweight (BMI <18.5 kg/m2).
- Medication is not helping reach glycaemic or weight targets and is not being prescribed for cardiovascular benefits.
The seven treatment pathways
Additional advice: GLP-1 receptor agonists and tirzepatide
● Currently recommended GLP-1 RAs are liraglutide, dulaglutide or oral semaglutide (at any licensed dose for type 2 diabetes), and subcutaneous semaglutide (up to 1 mg).
● Stop GLP-1 RAs or tirzepatide:
- If the person becomes underweight (BMI <18.5 kg/m2).
- If the medication is not helping reach glycaemic targets and is not being prescribed for cardiovascular benefits.
● Share MHRA advice on use of GLP-1-based medicines in pregnancy and breastfeeding with all people of childbearing potential.
● Share MHRA advice on GLP-1-based therapies and risk of severe acute pancreatitis.
● For guidance on preventing negative impact on eyes, see the Effects of rapid reduction in HbA1c section of the NICE NG242 guideline on diabetic retinopathy, and the MHRA guidance on non-arteritic anterior ischaemic optic neuropathy, both summarised below:
- When starting treatment likely to result in rapid, substantial HbA1c reductions, notify the person’s ophthalmologist (if the person is attending specialist hospital eye service) so they can assess eyes before treatment begins if they feel this is needed.
- Non-arteritis anterior ischaemic optic neuropathy (NAION), which can cause sudden deterioration in vision, usually in one eye at a time, has been very rarely reported in association with semaglutide use for type 2 diabetes, weight management and cardiovascular risk reduction. Patients reporting a sudden loss of vision (including partial loss) while on semaglutide treatment should be urgently referred for ophthalmological examination. People taking semaglutide should be warned to seek urgent ophthalmological assessment if they develop any visual loss.
- Refer to an ophthalmologist in accordance with the NHS Diabetic eye screening pathway standards – this will usually be undertaken by the retinal screening service.
Changes to insulin therapy guidance
● Provide support from a healthcare professional trained in insulin therapy.
● Discuss risks and benefits of insulin, and benefits of continuing medications that improve cardiorenal outcomes and weight loss.
● Offer once-daily or twice-daily basal insulin as initial therapy. Consider combining basal and short- or rapid-acting (or pre-mixed) insulin if HbA1c is ≥75 mmol/mol.
● Consider pre-mixed regimens containing insulin analogues if preference is to inject just before meals, or if there are hypoglycaemia problems or a rapid rise in blood glucose after meals.
● If multiple basal insulins are equally suitable, use the least expensive option.
Rationale and impact of the updated guidance
Metformin M/R versus standard-release
● Similar effectiveness; safety for hypoglycaemia; lower risk of gastrointestinal adverse events; may have better adherence; may cost less than standard-release (as of December 2025).
● Standard-release may be preferable for those with swallowing difficulties, as tablets can be crushed or a liquid formulation may be used. This may be appropriate for people with dementia or learning difficulties, where dysphagia is more common.
● Those tolerating standard-release metformin can continue unless the person prefers to switch.
Choice of SGLT2 inhibitors
● NICE commented on the large price reduction of generic dapagliflozin but chose not to specifically recommend this, as other similarly effective SGLT2is may become cheaper in the future. While dapagliflozin is the least expensive of the suitable SGLT2is, NICE supports its use because it is likely to reduce the cost of implementing the recommendations without negatively affecting quality of care.
Subcutaneous semaglutide (up to 1 mg per week) for people with ASCVD
● Best evidence for cost-effectiveness in people with ASCVD.
● Likely to increase immediate costs but reduce DPP-4 inhibitor use.
● Increased weight loss may result in better long-term prognoses and, if maintained, will reduce needs for long-term and later-stage treatments (e.g. insulin).
● Liraglutide will have a lower price when generic but is not sufficiently clinically effective.
● Evidence on tirzepatide and oral semaglutide for ASCVD was not available at time of guideline review.
● Clinicians are referred to MHRA guidance on potential side effects of incretin-based agents and pancreatitis, and on semaglutide and non-arteritis anterior ischemic optic neuropathy.
Early-onset type 2 diabetes (<40 years)
● Consider GLP-1 RAs and tirzepatide along with metformin M/R and SGLT2is in people with early-onset type 2 diabetes, as this is a relatively small group and health inequalities are likely if they do not receive treatment early.
● Although there is no clinical trial evidence supporting use of GLP-1 RAs and tirzepatide specifically in this group, they were recommended to reduce the very high lifetime risk of cardiovascular complications.
GLP-1 RAs and tirzepatide for people with obesity
● These drugs were not cost-effective in economic modelling for this population, as lifetime CVD risk is lower than in those with early-onset type 2 diabetes, so they were not recommended as first-line therapy. However, they can be considered if further treatment is needed to achieve individualised glycaemic targets.
● This advice is specifically for managing type 2 diabetes. If considering medication primarily for weight management, follow NICE NG246 guidance on overweight and obesity management.
Chronic kidney disease
● Although the FLOW trial has identified clinically important benefits for renal protection and glycaemia (N Engl J Med 2024;391:109–12), subcutaneous semaglutide was not cost-effective for this population in the economic model, hence GLP-1 RAs or tirzepatide were not recommended.
● Since SGLT2is will continue to have renal and cardiovascular benefits but have reduced or absent glucose-lowering effects at eGFR below 45 mL/min/1.73 m2, they should be continued but a DPP-4i may also be needed.
Biosimilar insulins
● Biosimilar insulins may offer considerable cost savings.
● To gain approval, they have to be as safe and effective as the original reference medicine, so for new initiations, when a biosimilar is available, it is appropriate to use the one at lowest cost.
● Consider carefully planned and shared decision-making around switching to a biosimilar insulin at routine reviews.
Linked and other useful resources
NICE NG28 updated guideline
- NG28: Type 2 diabetes in adults: management
- Visual summary of medicines recommendations
- Visual summaries: Choosing, reviewing and changing medicines and choosing medicines for first-line and further treatment
- Patient decision aid: HbA1c targets
Other NICE guidelines
- NG246: Overweight and obesity management
- NG56: Multimorbidity: clinical assessment and management
- NG238: Cardiovascular disease: risk assessment and reduction
- NG106: Chronic heart failure in adults: diagnosis and management
- NG203: Chronic kidney disease: assessment and management
- NG17: Type 1 diabetes in adults (Initial diagnosis section)
- NG242: Diabetic retinopathy: management and monitoring
NHS resources
- Type 2 Diabetes Path to Remission Programme
- NHS Better Health – dietary guidance
- Language matters: language and diabetes
- NHS Diabetes eye screening pathway





Introducing the PCDO Society’s new podcast. In episode 1, we cover the updated NICE NG28 guideline on type 2 diabetes management.
24 Feb 2026