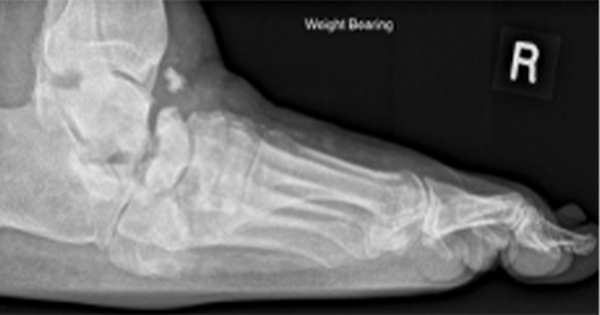
Over the last 25 years, one of the most significant, and potentially most controversial, areas of clinical development has been in the area of wound dressings.
Here, David Wylie, co-chair of the Foot in Diabetes UK (FDUK), invited six members of the FDUK executive committee to respond to six questions about wound dressings — with specific reference to silver dressings with this series being linked to the 25th ‘silver’ anniversary of the journal!
Here, he synthesises their responses to provide an overview of the wound dressings landscape and the role played by dressings in a constantly changing and rapidly developing clinical environment.
1) What have been the most significant developments in dressings over the last 25 years?
It is evident that there has been a paradigm shift in wound management within the foot and lower limb over the last 25 years. Much of the early research into this emerged from the nursing profession. This was driven initially by the move to maintaining a moist wound environment to facilitate healing, the evidence base for which goes back as far as the early 1960s (Winter, 1962; Hinman and Maibach, 1963). Non-adherence became (and remains) the foundational enabling the principle of moist wound healing (Korting et al, 2011).
This evidence drove the need for a generation of wound dressings that would support this principle. Currently, there are upwards of 5,000 wound care products supporting the moist wound-healing environment (Shah, 2011) containing a range of highly absorbent materials, including alginate, foam or carboxymethylcellulose. There is also a wide range of occlusive, non-occlusive and semi-occlusive dressings into which these elements may be incorporated. Additionally, bioengineered tissue, negative pressure and hyperbaric oxygen therapy are all available as wound management options.
From this bewildering array, the vacuum-assisted closure (V.A.C) pump has been one of the greatest advances in foot wound closure — particularly in post-surgical wounds — as recommended by the International Working Group on the Diabetic Foot (Schaper et al, 2019) which states that clinicians should: “Consider the use of negative pressure wound therapy to reduce wound size, in addition to best standard of care, in patients with diabetes and a postoperative (surgical) wound on the foot.”
More recently, there was the publication of robust evidence to support the efficacy of a lipocolliod technology dressing impregnated with nano-oligosaccaride factor in the management of neuroischaemic diabetic foot ulceration in 2013 (Shanahan, 2013). This double-blind randomised controlled trial changed the paradigm by which wound dressings were evaluated.
Gone are the days of using a product because it has always been used. Evidence is now being accumulated to demonstrate from observational studies like Münter et al (2017) and Lázaro-Martínez et al (2019) that time to closure of diabetic foot ulcers and that to achieve 50% reduction of the Pressure Ulcer Scale for Healing pressure ulcers on the foot is substantially shorter when treated timeously with TLC-NOSF dressings as a first-line rather than second-line dressing.
This evidence based approach ought to provide enough impetus to support widespread change in clinical practice, however, as noted in one of the previous publications in this series, affecting such changes, even when clinical evidence appears compelling is not always easy (Shafaghat et al, 2021; Wylie, 2022) with confirmation bias and lack of leadership making it difficult to operationalise evidence on scale.
Indeed, although the NICE medical technologies guidance MGT42 (NICE, 2019) advocates UrgoStart for treating diabetic foot ulcers and leg ulcers by stating clearly that “healthcare professionals are expected to take this guidance fully into account”, it also acknowledges that any such guidance “does not override the individual responsibility of healthcare professionals to make decisions appropriate to the circumstances of the individual patient”. This irreconcilable dichotomy remains an insurmountable problem in the implementation of evidence-based practice.
Nonetheless, this evidence based approach has taught the podiatry profession that it can legitimately question the function and performance of wound dressings in a way that was not possible for the previous 25 years.
2) What has the last 25 years taught us about the importance of dressings by comparison with other wound management treatment modalities?
Traditionally, dressings were considered to provide only one element of the management strategies to support wound healing in the foot and lower limb, alongside pressure redistribution, debridement, vascular assessment, infection and debridement.
However, it is now evident that certain dressings can achieve increased healing compared with placebo using sucrose-octasulfate impregnated dressings. A total of 48% of neuroischaemic ulcers healed at 20 weeks vs 30% in the control group (P=0.002) and a significant reduction in time for the wound to close of 60 days (180 days vs 120 days, P=0.029) was also reported by Edmonds et al (2019).
This study also informed a series of best practice statements in the IWGDF Practical Guidelines (Schaper et al, 2019):
“Consider the use of the sucrose-octasulfate impregnated dressing as an adjunctive treatment, in addition to best standard of care, in non-infected, neuro-ischaemic diabetic foot ulcers that are difficult to heal.
“Consider the use of placental derived products as an adjunctive treatment, in addition to best standard of care, when the latter alone has failed to reduce the size of the wound.”
“Consider the use of autologous combined leucocyte, platelet and fibrin as an adjunctive treatment,in addition to best standard of care, in non-infected diabetic foot ulcers that are difficult to heal.”
So, although the need for a holistic approach involving infection control, good debridement of devitalised tissue, diabetes control and pressure reduction remains vitally important, it may well be that dressing choice, especially in the early stages of the wound healing process, is assuming a more prominent role.
3) Why did silver dressings become so popular?
Wound care practitioners are always concerned with the development of infection, and silver dressings came to be regarded as efficient in reducing bioburden in colonised and infected wounds. They were thought also to decrease the risk of infection in burns and skin grafts.
When silver came to market, there was a gap in available dressings with options historically being limited to iodine and cholorhexidine-based dressings. Silver provided a new and exciting option in treating what came to be described as critical colonisation (now known as covert infection) — particularly in leg ulceration. Clinicians began to understand the ‘tipping point’ theory and began to utilise topical antimicrobials on wounds ‘just in case’.
The introduction of silver, therefore, coincided with the wound care paradigm of “contamination, colonisation and infection” to which these dressings seemed particularly relevant.
This clinical — but non evidenced — practice led to overuse of topical antimicrobial silver dressings, and led to a proliferation of silver-based wound care products and dressings. However, this approach is now deemed on reflection to have ultimately generated an unhelpful, often unthinking explosion in their use.
4) What were the strengths and challenges of the silver revolution?
Strengths
The primary strength was that it brought the paradigm of “contamination, colonisation and infection” into the public domain and increased the general awareness of infection. They were also reasonably priced, had few allergies, were easy to apply and could be left in situ for several days.
Challenges
Although silver has been proven, in vitro, to effectively kill the bacteria present in most wounds (or at least help prevent critically colonised wounds becoming infected) some challenges remain in justifying its indiscriminate utilisation.
These were eloquently summarised in by Bergin and Wraight (2006) in a Cochrane Systematic Review Systematic which concluded that “despite the widespread use of dressings and topical agents containing silver for the treatment of diabetic foot ulcers, no randomised trials or controlled clinical trials exist that evaluate their clinical effectiveness. Trials are needed to determine clinical and cost-effectiveness and long term outcomes including adverse events.”
The scepticism surrounding silver dressings was augmented by Michaels et al (2009) in the VULCAN trial, which concluded: “There was no evidence to support the routine use of silver-donating dressings beneath compression for venous ulceration.”
These caveats and cautions, together with increased scrutiny on antimicrobial and antibiotic stewardship provided a necessary clinical corrective to the injudicious use of silver dressings and helped clinicians to use their critical clinical decision making skills when utilising them.
5) Where does silver sit now in the wound management therapeutic armoury?
Integrating silver-based dressings in the wound care ‘ladder’ remains one of the ongoing challenges for clinicians across the whole spectrum of wound care. Finding the best point on the wound management pathway to utilise silver requires good clinical assessment skills and an understanding of its advantages and limitations.
Helpfully, a recent review by Khansa et al (2019) concluded that “in infected wounds, silver is beneficial for the first few days/weeks, after which non-silver dressings should be used instead. For clean wounds and closed surgical incisions, silver confers no benefit”. This provides some guidance to assist clinical decision making in relation to the indications for utilising silver dressings, and — equally importantly — a helpful caution as to when to refrain from their use.
6) What part do topical dressings have to play in the future of wound care, especially now that podiatrists have increased access to medicines through POM-S and prescribing?
Dressings, particularly those that have been through the trial process will continue to be used. However, there is a global drive, possibly linked to sustainability, to use simpler, less expensive, non-adherent dressings that can be removed more frequently to allow more frequent assessment and debridement.
This, together with improved systemic infection control using oral antibiotics and improved glycaemic control, is likely to provide the future paradigm of wound management for the foreseeable future rather than being seduced by the siren song of the latest fad in expensive dressings with a limited evidence base.
The main challenge across the wound management spectrum, therefore, remains a lack of robust evidence. Even where evidence does exist, overcoming confirmation bias, custom and practice and individual clinician preferences will remain difficult in supporting change in the area of wound dressing behaviours.