Over the past few decades, the incidence of diabetes has consistently increased globally. The World Health Organization (WHO; 2023) estimates that 422 million people are currently living with diabetes; a 48% increase from 2010. This upward trajectory is reflected in the findings of the 2021 Scottish Diabetes Survey, which reported that 329,927 people in Scotland have been diagnosed with diabetes (NHS Scotland, 2021). This is 6% of the population, and is a 1.4% increase from 2010, when the survey reported 237,468 people with diabetes.
This rise has also led to an increased number of people living with complications of diabetes, including nephropathy, vascular disease and neuropathy. When these complications develop in the foot, along with potential structural deformities, there is an elevated risk of infected ulceration and/or Charcot neuroarthropathy (CN; Lees et al 2006; Crawford et al, 2020). These conditions may be present individually, but where they have a potential to co-exist, differential diagnosis is a significant challenge in a clinical environment as both produce clinical signs of erythema, rubor and oedema (Womack, 2017; Dardari, 2020).
A systematic approach to clinical history taking and physical examination of the foot can help to identify the correct diagnosis, but additional diagnostic tests may be required to make a definitive decision.
Diagnostic imaging
CN is a chronic, devastating and destructive disease of the bone structure and joints in patients with peripheral neuropathy. Although it is a relatively rare complication of diabetes, with a prevalence of 0.08-0.4%; without early intervention to immobilise the affected joints, CN can cause significant deformity of the natural foot architecture, making the foot more vulnerable to ulceration and potential amputation. (Womack, 2017; Lever, 2018; Dardari, 2020). In its acute phase, the condition presents as a red, hot, swollen foot and in the presence of peripheral neuropathy, may be painless. These characteristics can also be observed in the more common presentation of osteomyelitis in the neuropathic, diabetic foot, which makes it challenging to differentiate between the two conditions.
For diagnostic imaging, plain film radiography is recommended initially for both conditions, with serial imaging being useful for monitoring disease progression. Radiographic features of osteomyelitis include periosteal reaction, cortical destruction, bone lysis and patchy sclerosis. However, the same radiographic features may also be observed in CN, with the addition of fractures and joint subluxation as the condition progresses (Womack, 2017; Lever, 2018; Bus et al, 2023). These overlapping signs greatly affect the ability of radiography to differentiate between CN and osteomyelitis, with studies estimating that it is truly diagnostic in only 50–60% of cases (Lipsky et al, 2004). The presence of demineralisation, periosteal reaction and cortical destruction are seen as being the most diagnostic features (Hartemann-Heurtier and Senneville, 2008).
Magnetic resonance imaging (MRI) is recognised as being more sensitive and specific than plain film radiographs in assessing diabetic foot disease. While both CN and osteomyelitis produce low signals on T1 sequencing, T2-weighted images may be of more value.
In CN, a lower signal is anticipated, while osteomyelitis produces a high signal (Marcus et al, 1996). A retrospective review by La Fontaine et al (2021) comparing MRI results with bone biopsy in the diagnosis of osteomyelitis found that 29.3% of their cohort had been misdiagnosed based on preliminary radiology reporting. A subsequent review of the MRI images for research purposes reduced this error margin to 16%, but the specificity rate remained at 74%, with a sensitivity of 87%. It was noted that the presence of reactive bone marrow oedema could potentially lead to diagnostic inaccuracy, as this is also evident with acute CN, trauma and other inflammatory bone diseases (La Fontaine et al, 2021). These findings were supported by a systematic review of the literature in 2023 (Wukich et al, 2023).
Infrared thermography
The infrared (IR) spectrum is made up of wavelengths ranging from 0.75 to 1,000 µm. However, the human body emits a much narrower range of IR or thermal radiation, specifically wavelengths ranging from 8 to 12 µm. This radiation can be measured for diagnostic purposes via thermal imaging cameras which detect variations in skin temperature. The captured data is then used to generate a visual map of temperature distribution across the skin by a graded colour scale. This technology has several potential applications in medicine, including monitoring blood flow, detecting breast cancer and assessing muscular performance in the human body (Bagavathiappan et al, 2010). This technology provides a safe, non-invasive imaging technique without the production of ionising radiation.
Thermal imaging can be a valuable tool in the management of diabetic foot disease. It allows for early detection of pre-ulcerative tissues, assessment of blood flow, monitoring healing progress and facilitates prevention and patient education. Handheld infrared temperature measurement devices have been increasingly common in identifying and monitoring ‘hot spots’ within the diabetic foot. Research addressing preventative measures suggests that daily temperature monitoring may provide an appropriate solution to pre-empting ulcer formation, as an increased temperature at a localised skin area is believed to precede the development of an ulcer.
Bus et al (2021) demonstrated the potential for at-home temperature monitoring in a randomised controlled trial. Using handheld infrared thermometer participants of the intervention cohort were asked to measure 6–8 points of the plantar aspect of both feet daily. Results of the study showed at-home temperature monitoring reduced the incidence of ulcer reoccurrence by 22% compared to that of standard care. This result was considered to be due to the participant reducing ambulatory activity after identifying a hot spot. Limitations of the study include the handheld method of measurement. It is difficult to confirm that the same points were consistently captured. In addition, there is potential for inflammation, with the measured areas to be missed.
Results from a study by Lavery et al (2004)showed similar potential to at-home temperature monitoring. Using a handheld infrared skin thermometer (TempTouch; Xilas Medical) participants were asked to monitor six predetermined sites on the plantar aspect of the feet both morning and night. The results of the pilot study suggested temperature monitoring may be an effective means of identifying early signs of foot ulceration.
The handheld method of temperature monitoring was a limitation of both studies, for both reliability of results due to lack of consistency in measurement areas and potential areas missed (Lavery et al, 2004; Bus et al, 2021). Newer technology allowing temperatures to be measured across the whole foot in a single image may overcome these issues. The recently published guidance from the International Working Group on the Diabetes Foot recommends those who are deemed to be at moderate or high risk of foot ulceration be encouraged to use daily skin temperature monitoring to identify any early signs of foot inflammation and help prevent a first or recurrent plantar foot ulcer (Bus et al, 2023). In addition, the guideline suggests that temperature measurement may be used to monitor Charcot neuroarthropathy, although there are no current recommendations for this to be carried out via thermal imaging rather than handheld infrared thermometers.
Case study
A 67-year-old woman presented to podiatry with a new episode of ulceration affecting the left fifth metatarsophalangeal joint. This had been a recurrent wound site despite regular podiatry input for callus reduction and the provision of orthopaedic footwear with custom insoles. She had well-controlled type 1 diabetes, with an HbA1c of 45mmol/mol. Additional comorbidities include chronic kidney disease (stage 3) and heart failure. Past foot health history includes amputation of the right second to fifth toes secondary to infection. Although unilateral disease is more common, she had previously had active Charcot neuroarthropathy affecting both feet, with the right foot being affected in 2009 and a subsequent episode affecting the left foot in 2018. Examination of the feet revealed palpable, biphasic pulses and peripheral neuropathy with a score of 0/10 on 10 g monofilament assessment.
At first presentation, the wound was debrided and appropriately dressed. She was initially supplied with a temporary pressure redistribution shoe with a rocker sole. A custom insole was manufactured and fitted to this shoe at the earliest opportunity via the multidisciplinary diabetes foot clinic (MDFC).
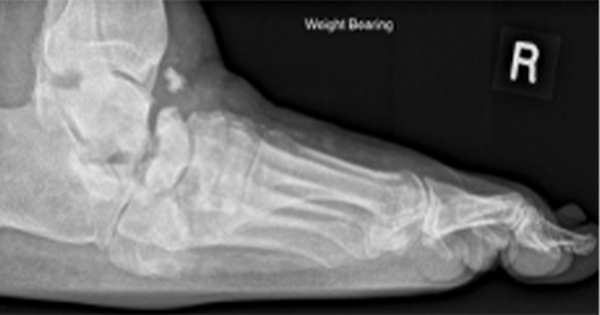
Two months after presentation, the patient presented with clinical features of new infection to the wound. Wound swabs identified group G Streptococcus with sensitivity to penicillin; therefore, an initial 7-day course of phenoxymethylpenicillin was prescribed. Although this improved the clinical signs of infection, it did not fully resolve it. An X-ray was requested to exclude underlying osteomyelitis and revealed cortical destruction at the head of the left fifth metatarsal, compatible with osteomyelitis. It also generated an incidental finding of degenerative changes affecting the mid-foot joints (Figure 1).
It was unclear from the imaging if the bony changes represented an active infection or old changes from the previous episode of osteomyelitis. An MRI was therefore requested to confirm, and osteomyelitis treatment was initiated.
The wound continued to reduce in size and clinically improved. However, when the MRI scan was performed 4 weeks later, the radiology report identified advanced arthropathy within the tarsometatarsal joints with degeneration, bony fragmentation, joint space loss and bony oedema in keeping with Charcot’s arthropathy (Figure 2). Both the patient and the podiatrist who reviewed the MRI were concerned regarding the possibility of Charcot recurrence, as the report did not specify if the condition was quiescent. Temperatures checked with a handheld thermometer (Optris LS Infrared Thermometer) at the first tarsometatarsal joint (TMTJ) demonstrated a 5.4° temperature difference between the feet suggesting possible reactivation of CN within the midfoot joints. The patient was therefore supplied with a rebound walker boot to immobilise and offload the foot, and the patient was referred urgently to the MDFC for review.
On review at the MDFC, minimal erythema was noted at the periphery of the ulcer on the left fifth metatarsophalangeal joint. This had improved significantly since antibiotics were initiated and appeared to be localised to the wound periphery. Assessment with a hand-held temperature gun demonstrated a 2.9° temperature elevation at the left first TMTJ when compared with the contralateral limb (points A and B on Figure 3). Thermal images were taken using the ThIR-A615 Infrared Thermal Imaging System (Thermidas Oy) to evaluate the distribution of temperature across the feet (Figure 3). This device captures images at a resolution of 640 × 480 pixels. Although there was still an ongoing temperature difference between the feet at the first TMTJ, it can be clearly seen that the temperature is radiating from the wound site with a 7° temperature difference identified between points C and D on Figure 3 and does not originate at the medial midfoot joints. This was not evident on clinical examination which showed minimal inflammation.
Recent X-rays were compared to older images that showed no progression of bony deformity. The team agreed that the temperature change was representative of ongoing osteomyelitis and not new Charcot. This could be clearly demonstrated to the patient using the thermal images, providing reassurance and increasing her confidence in resuming her Darco shoe with custom insole. She found the Darco device more comfortable and easier to cope with. The patient completed the course of antibiotics. Temperatures across the foot stabilised within 2 weeks and the wound went on to fully heal.
Discussion
The ThIR-A615 Infrared Thermal Imaging System allowed the team to assess temperature fluctuation across the whole foot thus clarifying the origin of the temperature rise. This was not possible with the handheld device that is more routinely utilised. The use of thermal imaging in this case facilitated the early transition from a knee-high rebound walker to a lighter weight temporary shoe. Without a clear diagnosis, this level of immobilisation would have continued until temperatures had been measured at less than 2° difference between the two feet for a minimum of 6 weeks, as per local guidance. This had a significant positive impact on the patient’s quality of life allowing her to maintain a greater degree of independence.
Thermal images can play a valuable role in patient education and allow the individual to gain a clearer understanding of their condition. In this case, the visual representation of inflammation within the foot assisted the team in having a balanced conversation with the patient, reducing her anxiety with regards to CN recurrence, and providing reassurance on the origin of the raised temperature within her foot. The patient was then able to make an informed choice about her treatment plan.
While thermal imaging has demonstrated its potential within the foot screening arena, its place within the management of active diabetes foot disease is unclear. In this case, thermography did aid clinicians in establishing a clear diagnosis and impacted on the clinical management plan. It is, however, important to note that it is was used in conjunction with other diagnostic methods for a comprehensive evaluation. Further research and clinical studies are continually being conducted to strengthen the evidence and refine the use of thermal imaging in the care of the diabetic foot.