Diabetic foot disease is one of the most common and feared complications of diabetes. Foot ulceration affects 5% of patients with diabetes and precedes a lower-limb amputation in diabetes in 80% of cases. Prevention and healing of diabetic foot ulcers is, therefore, critical in preventing lower-limb amputations and maintaining quality of life for the patient (Ramsey et al, 1999).
Larval therapy is used worldwide to heal chronic and complicated wounds, including vascular, neuropathic and pressure ulcers, and also to treat trauma-related injuries, such as burns and surgical wounds. The following case report highlights the use of larvae therapy in a patient with a chronic non-healing diabetic foot ulcer.
Case history
A 61-year-old female with a 30-year history of type 2 diabetes on insulin presented to the diabetic foot clinic with an infected neuropathic intra-digital ulcer. The ulcer was between her left fourth and fifth toes with a surrounding cellulitis spreading up the left leg. Her diabetes had always been difficult to control and was complicated by diabetic proliferative retinopathy, renal impairment, neuropathy, hypertension and high cholesterol. Her HbA1c around the time of presentation was 78 mmol/mol (9.3%).
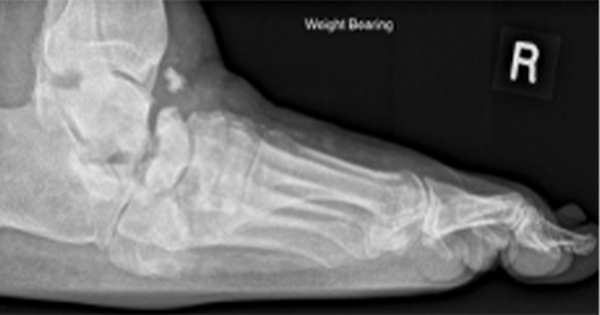
Investigations showed a normal white cell count of 6.4×109/L, neutrophils of 3.2/uL, raised erythrocyte sedimentation rate (ESR) of 40 and C-reactive protein (CRP) of 140 mg/L. Her deep wound swab was positive for Extended Spectrum Beta Lactamase (ESBL) and Proteus. An X-ray of the left foot showed marked swelling of the overlying soft tissues with bony resorption of the head and proximal shaft of left fourth and fifth metatarsals. Her MRI foot confirmed the presence of osteomyelitis in her left fourth and fifth toes. Her Ankle Brachial Pressure Index showed a value of 1.4 on the right and 0.95 on the left consistent with non-compressible vessels due to excessive vascular calcification. Toe Branchial Index of the right and left limbs were, however, both normal with values of 0.82 and 0.81, respectively.
Treatment consisted of offloading the foot, intravenous Piperacillin/Tazobactam for 6 weeks, improved blood glucose control and regular debridement of necrotic tissue. Numerous dressings were applied to the ulcer with little benefit. One of the difficulties in dressing application was the location of the ulcer in between the toes on the sole of the foot.
After 2 to 3 weeks, little progress was made and lower-limb amputation was being considered. However, as a final option, bio-therapy in the form of larvae treatment was tried. The larvae were ordered from the UK and, with the consent of the patient, the larvae were placed on the foot ulcer in a sterile BioBag® (BioMonde). A hydrocolloid dressing, which consisted of a combination of gel-forming polymers to help as an absorbent to manage wound exudates by swelling into a gel-like mass, was applied at the edge of the foot ulcer to prevent excoriation of the surrounding skin. The bio bag was then covered with a sterile saline-moistened gauze swab to prevent the larvae from becoming dehydrated and inactive. The dressings were checked daily to ensure that the larvae were still alive. One bio bag dressing was left in place for a maximum of up to 4 days. As the maggots finish secreting their proteolytic enzymes in up to 72 hours, the wound bed was closely reassessed at every dressing change for a change in colour of the eschar. The old dressing was removed if the larvae had finished secreting the enzymes.
Wound debris can harbour bacteria rendering it to a risk of infection. Therefore, the need for a next treatment cycle was determined regularly depending on the extent of the remaining necrotic tissue. Significant de-sloughing and heavy exudate around the ulcer were observed in between the treatment cycles. Due to the extensive nature of her foot ulcer, the patient required three sets of applications, which remains the average amount of applications at most treatment centres. During the following few weeks, the ulcer started to show a significant clinical improvement. Healthy granulation tissue and reduction in wound size became progressively evident during follow-up visits. Finally, over a period of 5 months, the ulcer healed completely and the patient was back mobilising fully. Photographs prior to and post application were taken and kept for records (Figure 1).
Discussion
Achieving ulcer healing in diabetes is difficult. Most patients with complicated foot ulcers require an inpatient stay and in Ireland alone, the inpatient cost of treating a diabetic ulcer over 1 year was estimated at €23,500 per patient (Smith et al, 2004). Failure to treat can be catastrophic and lead to foot amputation. However the International Diabetes Federation has established that amputation risk can be decreased by 49% to 85% with the implementation of appropriate care strategies (International Diabetes Federation, 2012).
The management of a foot ulcer includes multidisciplinary input, intensive diabetes control, offloading, improving the vascular supply to the affected foot and ensuring the ulcer is not infected, regular debridement and allowing the tissue to heal in a sterile environment conducive to tissue healing and wound regeneration (Kavitha et al, 2014).
Larval therapy or bio surgery has been used to assist in wound debridement and healing for decades (Baer, 1931). However, it was not used to its full potential in the past. Clinical trial data and meta-analyses since 1990 have reported the efficacy and safety of larvae therapy in comparison with more conventional and invasive treatments, including surgery in the diabetic population (Mumcuoglu et al, 1998; Armstrong et al, 2005; Tian et al, 2013). It is, however, often used as a last resort treatment for wounds where conservative measures fail and surgery is not considered due to other medical co-morbidities.
Larvae are sterile maggots of the common green bottle fly, Lucilia sericata. They not only physically feed on the necrosed tissue, but they also exhibit a dual mechanical and secretory effect on the wound (Nigam et al, 2006). First of all their movement helps disintegrate the sloughed tissue. Secondly, the larvae secrete proteolytic enzymes, such as trypsin and chymotrypsin, to liquidise necrosed tissue, which they subsequently ingest (Choudhary et al, 2016).
Evidence also suggests that larval secretions are antimicrobial and, therefore, combat wound infections by substantial destruction of wound pathogens reducing the bioburden within the wound. Wound bacteria often aggregate to form a resistant layer known as a biofilm. This is extremely difficult to eradicate with antimicrobials or topical applications and, therefore, add to chronicity of the wound and prevent ulcer healing. Larval secretions have been shown to disrupt this pre-formed biofilm and preclude further formation (Sherman, 2014). The larvae also appear to have an effect on promoting fibroblast proliferation and collagen synthesis within the wound, which promote wound healing (Horobin et al, 2006). The effects of larval secretions on inflammatory mediators, such as neutrophil, moncocyte and macrophage function, have been investigated and it is now recognised that secretions from the larvae blunt the effects of these mediators and thus attenuate a chronic inflammatory response in the wound. The larval secretions have a particular effect on neutrophil function, not only reduce production and release of tissue damaging substances, such as elastase and hydrogen peroxide, but also inhibiting chemotaxis of neutrophils into the infected site (Horobin et al, 2006).
There has been a noticeable increase in the use of larvae therapy over recent few years (Strohal et al, 2013). This form of bio surgery has successfully been used to avoid lower-limb amputation in some patients with complicated and compromised diabetic foot ulcers (Sherman, 2009). Hence, it is now seen as a safe and effective treatment.
Conclusion
This case report demonstrates the holistic approach of a multidisciplinary team towards the management of a diabetic foot ulcer. Larva therapy allowed the skin to reepithelialise and heal, and reduced the length of the hospital admission. Scientific evidence supporting the efficacy and safety of larvae therapy is growing. However, there still exists the need to conduct large prospective clinical trials. This necessitates exploring the mechanisms and developing a complete understanding of this interesting wound-healing technique.