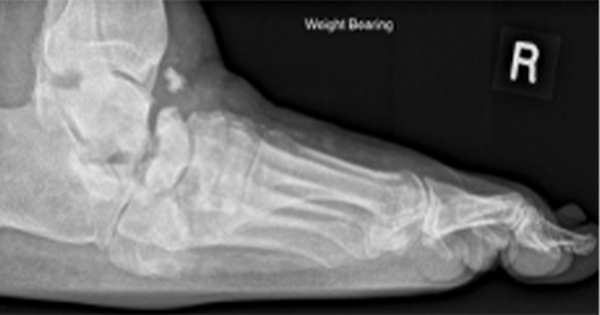
Ulceration of the foot remains one of the most feared complications of diabetes mellitus, and has been found to affect approximately 15% of all patients with diabetes at some stage in their life (Palumbo and Melto, 1985; Reiber, 1996). Each ulcer carries with it the possibility of months or years of incapacity, as well as the threat of the loss of a limb. The mean annual incidence of lower limb amputation for diabetes in Westernised countries is in the order of 10 per 100 000 population (Anon, 1997). The costs to healthcare systems, social services and to individuals are enormous (Apelqvist et al, 1995).
The management of individual ulcers is based on the art of doctors, nurses and podiatrists, and this in turn is based as much on experience and belief as on objective evidence. Although the magnitude of the problem is acknowledged, there have been remarkably few controlled studies of either prevention or treatment. One reason for this is the absence of any system for classifying ulcers (LeFrock and Joseph, 1995).
Since foot lesions are of multiple different types, with different causes and prognoses, it follows that the selection of any particular ulcer for study requires the existence of an unambiguous system of classification. As no such system exists, it has been impossible to undertake scientific evaluation of different management strategies.
Early systems of classification
Although there is no classification system in widespread use, several have been proposed. These have been reviewed elsewhere (Jeffcoate et al, 1993; Lavery et al, 1996) and, in general, are either too simple to be specific, or too complicated to be remembered.
The San Antonio group has proposed two new systems: one that attempts to categorise both the ‘at risk’ (non-ulcerated) foot together with a protocol for care for established ulcers, and a second that is concerned solely with ulcer type (Lavery et al, 1996; Armstrong et al 1996). Both are linked to a series of strategies for clinical management, and to anticipated outcome. The second system has recently been evaluated (Armstrong et al, 1998).
Problems in devising systems of classification
There are a number of reasons why it has proved difficult to devise a classification system:
- The multiple factors that contribute to the development, or failure to heal, of different lesions.
- The difficulty in deciding which of these multiple aetiological factors predominates in any particular case.
- The lack of simple and reproducible methods for determining the presence, severity or extent of aetiological factors such as ischaemia, neuropathy (motor, sensory or vasomotor) and infection.
- There may be multiple ulcers, and different lesions on the same foot may be of different types. The outcome of one may be determined by the outcome of another.
- Ulcers change with time, and may either improve or deteriorate.
- People change with time, and many will develop recurrent ulceration over a period of years.
- There is no agreed glossary of terms used in the description of ulcers, with consequent lack of clear distinction between words such as slough and exudate, or eschar and gangrene.
Classifications vs descriptions
Another difficulty is failure to agree the purpose of the classification; yet this is crucial to the structure of the system used. If it is to be employed in clinical practice to aid management of particular patients, its components, complexity and structure will be different from those used to identify a population of ulcers for recruitment to a multicentre study.
A classification that is used to prompt an approach to clinical management is essentially a description, and is likely to be more detailed but less widely applicable (Macfarlane and Jeffcoate, 1998). On the other hand, a classification should form the basis upon which observations can be made of an ulcer’s management and/or outcome, either in one centre or in several. It should allow scientific evaluation of disease processes and their care, and is primarily a tool for assessing service provision – rather than an aid to the management of any particular case.
Temporal aspects of a classification
A description is necessarily appropriate only to the time at which it is made, and will change over time. On the other hand, the relationship between a classification and time is more complex. Factors that might be considered as the basis for a classification, and which could themselves change with time, include the age of the patient, quality of glycaemic control, smoking habit, other complications of diabetes (particularly the degree of associated peripheral vascular disease), as well as factors relating to the ulcer itself, such as size and presence or absence of infection or gangrene. Hence, a problem arises in defining the point at which any particular lesion should be classified.
For example, a neuropathic ulcer on the tip of a toe may be clean when first seen, but may later become infected. The infection could then involve bone, causing osteomyelitis of the digit. If the digit is then amputated, the infection could still recur in an adjacent digit some months later. Such a sequence of events is not uncommon in clinical practice, but it is not clear how such a lesion should be classified.
If, however, the purpose of the classification is to identify types of lesion for scientific study, it follows that the lesion in the above example could be classified four times over: as a clean neuropathic ulcer; as an infected neuropathic ulcer; as osteomyelitis; and as recurrent osteomyelitis. If it were to be classified four times, then the sum of all the lesions classified in a certain unit would not be the same as the total number of lesions seen, and would exceed it greatly. Thus, systems designed for the purpose of studying methods of care must be different from those used for departmental record-keeping.
Requirements of a classification
A classification needs to be simple enough to be remembered, and yet precise enough to be useful. The terms used must be unequivocal, with words which have an agreed meaning – in order that similar lesions will be similarly classified in different centres (Lazarus et al, 1994). It also has to be widely accepted and widely adopted.
Structure of a classification
Any classification of diabetic foot ulcers into a number of defined groups must be based on key elements that help delineate one group from another. Classifications based on aetiology fail because the cause of many lesions is either unknown or multifactorial. A successful classification has to be based on all of those key elements that contribute to the nature of the lesion. It should not, however, be so complicated that it cannot be remembered.
Key elements of a classification
The San Antonio group has emphasised that an important feature is assessment of severity (Lavery et al, 1996). The group graded ulcers according to their depth, since this has been shown to be closely related to anticipated outcome (Reiber et al, 1992). Lesions graded in this way can be allocated to groups of approximately equivalent severity. We favour consideration of both depth and cross-sectional area separately (see below).
A classification, however, must also take account of three other aetiological factors – any or all of which may apply – that contribute to the status of the lesion: sepsis, ischaemia and neuropathy. These factors are variously associated with anticipated outcome, and also determine broad strategies for management.
Qualification of key elements
The key elements of the classification need to be subdivided according to the extent to which they contribute to the status of the lesion. The way in which this is done must to some extent be empirical, especially as subdivisions may be either quantitative or qualitative.
Area
The cross-sectional area of an ulcerated lesion is important. It can be estimated in routine practice by measuring the greatest diameter. For prospective research, however, it should be determined more accurately using, for example, sterile transparent sheets marked with a grid. The area can be graded as follows:
- 0 Skin intact
- 1 <1 cm2
- 2 1–3 cm2
- 3 >3 cm2
Depth
The following system (proposed by Lavery et al, 1996) is easily applicable in clinical practice and has also been validated (Armstrong, 1998):
- 0 Skin intact – either non-ulcerated or healed
- 1 Superficial – involving skin and subcutaneous tissues but not reachingto tendon, periosteum or joint capsule
- 2 Penetrating to tendon, periosteum or joint capsule
- 3 Involving bone or joint spaces
Sepsis
Infection is nearly always the consequence of ulceration, rather than its cause (the main exceptions being paronychia and tinea pedis). Its presence both impairs healing and worsens the lesion (Reiber et al, 1992; Caputo, 1994; Jeffcoate and Finch, 1994). Infection of soft tissues in the ischaemic foot may cause the onset of gangrene by triggering thrombosis in small arteries already narrowed by atherosclerosis. Infection of bone may be impossible to eradicate and often leads to surgery (Lew and Waldvogel, 1997). It follows that early effective deployment of antibiotics will save limbs and lives. Sepsis may be a secondary factor, but it is one that is potentially linked with effective intervention.
However, the diagnosis and categorisation of sepsis may be difficult. Isolation of bacteria from surface swabs is insufficient, because infection needs to be differentiated from harmless colonisation (Chanteleau et al, 1996). It has been argued that isolation of bacteria from deeper tissues may be similarly non-specific, and bacterial contamination of bone in a patient without apparent osteomyelitis has been demonstrated (Jeffcoate and Macfarlane, 1995). Conversely, the absence of bacterial isolates from a swab does not exclude infection since swabs need to be handled with great care if less fastidious organisms are to survive (Jeffcoate, 1987).
Other tests, including measurement of white cell count, C-reactive protein and body temperature, are insensitive in these circumstances, and so the diagnosis of sepsis is primarily clinical. Unfortunately, this too may be difficult because the signs may be masked by ischaemia (or mimicked by neuropathic vasodilatation). Also, the symptoms may be masked by denervation (or mimicked by painful peripheral neuropathy). Particular problems are encountered in the markedly ischaemic foot (in which the only clue to infection of a foot lesion may be the onset of localised pain) and in the exclusion of bone infection, especially when the bone structure is deformed by Charcot’s neuropathic osteoarthropathy. The suggestion that the presence of osteomyelitis can be reliably inferred if it is possible to ‘probe to bone’ (Grayson et al, 1995) has not been confirmed in the recent major study by Armstrong et al (1998). In this study, infection was identified by bone biopsy in 23.9% (86/360) of such cases, but not in a further 6.4% (23).
The final problem with subdividing sepsis for the purposes of classification is that the categories are discontinuous, and reflect qualitatively different types of infection. Nevertheless, these do roughly conform to a scale of increasingly poor prognosis, as follows:
- 0 No infection
- 1 Surface infection, indicated by slough or exudate, but without clinical suspicion of cellulitis or osteomyelitis; also tinea pedis
- 2 Cellulitis
- 3 Osteomyelitis
Ischaemia
Impairment of effective circulation is the single most important factor responsible for delayed healing in diabetic foot ulcers. It may be impaired as a result of atherosclerosis and narrowing of arteries and arterioles (macrovascular disease) or as a result of endothelial malfunction, capillary shutdown or arteriovenular shunting from vasomotor neuropathy (microvascular disease) (LoGerfo and Coffman, 1984; Flynn and Tooke, 1992; Flynn and Tooke, 1995). Thus, assessment of the degree of ischaemia is an essential part of any classification. Although the clinical signs are well defined, there is no agreed system for ranking them in order of severity.
The limitations of clinical criteria, such as loss of digital hair growth, capillary blanching of the elevated limb, and inter-observer variation in palpability of foot pulses, have been highlighted by others (e.g. de Heus-van Putten et al, 1996). Nevertheless, it is generally agreed that if both pulses are easily felt in a foot, it is very unlikely that there is significant macrovascular ischaemia. Conversely, when one or more pedal pulses are impalpable (in the non-oedematous foot), it is likely that there is some degree of ischaemia. Experienced observers will also recognise a constellation of clinical features which are associated with chronic reduction in tissue perfusion, including thinness, redness and shininess of the skin, slow-growing and atrophic nails, and a tendency to develop small scabs at points of pressure on the toes.
A number of techniques are available for the quantitative assessment of foot blood flow, but they also are open to criticism (Takolander and Rauwerda, 1995). A hand-held Doppler can be used to determine systolic pressures at the ankle, and the ratio of ankle pressure to brachial pressure (ankle-brachial index or ABI) is a reliable indicator of moderately or severely reduced large vessel flow (ABI <0.9; ankle systolic pressure <100mmHg). It is, however, a poor means of confirming that arterial flow is adequate – partly because arteriosclerosis in the calf vessels renders them resistant to compression in up to 30% of cases (Emanuele et al, 1981), and partly because ankle pressure may be a poor indication of tissue flow if the main obstruction lies distally. Determination of systolic pressure at the level of the toe may give a better indication (Apelqvist et al, 1989), but is not generally done – even in specialist centres.
All of these methods, both qualitative and quantitative, are essentially measures of blood pressure within larger vessels. Even if reproducible, a measure of pressure is not necessarily a useful measure of the effectiveness of blood flow. It may also give little indication of capillary flow and, more importantly capillary function. It is deficiencies in this – resulting from shunting, changes in pressure-flow characteristics or ineffectiveness of endothelial transfer of oxygen and nutrients (Flynn and Tooke, 1992; Flynn andTooke, 1995) – that are likely to be the main determinant of defective wound healing.
If abnormal capillary function is the hallmark of ischaemia, it can be argued that tissue perfusion may be abnormal, even in patients with pedal pulses that are easily felt, e.g. patients with classic neuropathic ulcers. Since the peripheral neuropathy in such cases is known to be associated with arteriovenular shunting, it is possible that there is also an element of tissue ischaemia – a phenomenon that has been called the ‘capillary steal syndrome’ (Ward, 1982) – and there is evidence that this is the case (Flynn et al, 1988).
The results of attempts to resolve these difficulties by using direct measures of tissue blood flow or tissue oxygenation have been disappointing. Assessment by toe and foot plethysmography, laser Doppler fluxometry and transcutaneous oxygen tension (TcPO2) is not in widespread use, because the methods are time consuming, involve the purchase of specialist equipment, and the results have large intra- and inter-individual variation (Apelqvist et al, 1989).
The San Antonio group did not include different degrees of ischaemia in their classification, but referred simply to its presence or absence, as indicated by one or more pulses being impalpable, an ABI<0.8 and (unspecified) clinical signs or other measures. However, we feel that it is important to grade the severity of any ischaemia present. When it is noted but not critical, it is unlikely that early referral for angiography/duplex ultrasonography would be considered in the majority of centres. When it is severe, urgent referral for a vascular opinion is usually indicated. For this reason, we suggest the following grading:
- 0 Both pulses easily felt in the foot, with no clinical features of tissue underperfusion
- 1 Diminution of both pulses, or absence of one, with signs suggestive of reduced tissue perfusion
- 2 Absence of both pulses, with signs suggestive of reduced tissue perfusion
- 3 Gangrene
Neuropathy
The contribution made to foot ulceration by denervation is multifaceted. Thus, motor neuropathy may lead to ulceration as a result of abnormal pressure loading, while distal symmetrical anaesthesia may mean that a person is unaware of foot damage that has occurred, and may allow the ulcer to worsen through neglect. In addition, neuropathic shunting and changes of pressure and flow characteristics (vasomotor neuropathy) could lead to abnormal capillary function (Flynn and Tooke, 1995). These may result in altered integrity of the skin, making it more liable to damage, and could also delay the healing process. Finally, the part played by ischaemic neuropathy in patients with established macrovascular disease (Flynn et al, 1988) must not be ignored.
Given that the contribution made by neuropathies to foot ulcers is so complex, it is not easy to devise a precise method of categorising nerve damage for the purposes of classification. In clinical practice it is usual to document the loss of peripheral sensation by a pinprick (or 10g monofilament), but there are no robust criteria for defining when sensation is normal, diminished or abnormal, or at which sites it should be tested. Electromechanical devices for measuring vibration perception threshold (VPT) are of value for documenting severe established sensory neuropathy (Hoeldtke et al, 1994), but have too much inherent inter-rater variability for determining degrees of partial sensory loss. Since the measures of sensory neuropathy are imperfect and there are no measures at all for motor neuropathy in the foot, or for vasomotor neuropathy, it follows that any classification has to be primarily qualitative, and descriptive.
- 0 No detectable neuropathy: intact pinprick/monofilament/VPT sensation
- 1 Neuropathy contributory to the ulcer: impairment (reduced or absent) of pinprick/monofilament sensation/some elevation in VPT
- 2 Neuropathy as dominant factor contributing to the ulcer: absent pinprick sensation, with palpable pedal pulses
- 3 Charcot’s neuropathic osteoarthropathy
These categories do not form a logical progression, and do not simply reflect increasing severity of nerve damage, but they are mutually exclusive and, individually, are applicable to the vast majority of lesions encountered in clinical practice. Grade 2 includes the stipulation that the neuropathy which is a dominant aetiological factor should be associated with intact pedal pulses.
Charcot’s neuropathic osteoarthropathy is a distinct clinical entity, in which bone destruction within the foot is thought to result from a combination of sensory, motor and vasomotor neuropathies (Young et al, 1994; Brower and Allman, 1981; Young et al, 1995; Sanders and Frykberg, 1993; Jeffcoate et al, in press). Clinically, it is manifest either as an acute, tender inflammation associated with progressive distortion of the shape of the foot, or as a chronically misshapen foot which results in increased risk of ulceration from abnormal pressure loading.
The S(AD) SAD classification: size (area and depth); sepsis, arteriopathy, denervation
Incorporation of the above five elements allows any lesion to be classified – at any specified point. It allows them to be grouped according to (a) severity and (b) the dominant factors involved in their development. It is intended as an aid to audit and research, enabling certain types of ulcer to be identified for recruitment to prospective studies, as well as a means of comparing outcome between centres. It is not intended as a guide to management, and in this way differs from the San Antonio system (Lavery et al, 1996).
The S(AD) SAD system also differs from the San Antonio system in other ways:
- It categorises area as well as depth, whereas the San Antonio classification measures only depth. It remains to be proved that this adds specificity to the grading, but it seems likely that it will.
- It attempts to grade all five key components on a four-point scale, 0–3, with 3 being most severe. This is done without introducing greater complexity to the scheme (Table 1).
- It includes reference to neuropathy, which is omitted from the San Antonio system.
The adoption of a scheme such as the S(AD) SAD classification should allow selection of ulcers of all types for scientific evaluation of different management strategies – whether these involve investigation of the role of antibiotics, antiseptics, dressings – or of other interventions. It is only in this way that clinical practice will in future be based on a more scientific footing.
The remaining difficulty in any system of classification is agreeing at which point any one lesion should be classified. There are two broad options. For the purposes of audit of clinical practice, each ulcer should be classified only once, e.g. at the time of first referral. If, however, a classification is to be used to define populations of ulcers for recruitment into research projects, then it is conceivable that any one lesion could be classified on a number of different occasions as its condition improves or worsens – which was one intention of the classical coding described by Meggitt (1976) and Wagner (1981). The changing classification of the example mentioned earlier, the neuropathic ulcer that becomes complicated by osteomyelitis, is illustrated in Table 2.
Conclusion
The adoption of a robust classification is an essential requirement for undertaking prospective clinical research into ulcer management, which is so urgently needed. Although the San Antonio classification has been partially validated, it includes no reference to neuropathies. The current proposed classification attempts to encompass the most important factors that variously contribute to the presentation of any ulcer at any one time. It requires prospective validation to ensure that the suggested categories define groups of lesions which are mutually exclusive in a way that is clinically relevant.