Diabetic foot complications are common and vary considerably in both their pathophysiology and clinical presentations, ranging from subtle skin and joint changes through to full thickness ulceration and overt gangrene. A good healthcare professional must be able to interpret observations and have excellent methods of inquiry to ascertain a comprehensive medical history. It is, therefore, important that all those involved in screening, assessment, health education and treatment of people with diabetic foot problems are appropriately educated and trained, and are working within clearly defined guidelines and well supported infrastructures.
Encouraging patients to be responsible for their own diabetes, including basic foot care, is essential, but many patients are unable to monitor or inspect their own feet because of poor eyesight and/or reduced mobility (Thomson and Masson, 1992; Masson et al, 1989). Therefore, for the healthcare professional involved in foot care, maintaining regular contact with patients is important (Edmonds et al, 1996). Additionally, regular monitoring and screening by healthcare professionals provides an ideal opportunity to reinforce messages of self-care and its importance (Boulton and Malik, 1998).
Annual screening for neurological and vascular problems is the cornerstone of risk identification (Abbott et al, 1998; National Institute for Clinical Excellence [NICE], 2004); however, there are several other essential issues to consider when screening for the at-risk foot. These include: the presence of trauma-related callus, infections, limited and excessive joint mobility, skin and nail conditions, footwear, lack of self-awareness, poor healthcare beliefs/attitudes and limited access to appropriate foot care (Fletcher, 1990; Murray et al, 1996; Reiber et al, 1999; Uccioli et al, 1995). It is the aim of this article to cover those aspects related to skin and joints.
General appearance
A good habit to get into at the start of every appointment is to take time, stand back and make some general observations about the feet to be examined (Table 1). All of this information can be assimilated very quickly and should help confirm or refute the findings of assessment test results.
Skin and nails
The skin on the plantar surface of the feet is subject to high levels of repetitive shear, compressive and frictional forces during walking and standing. The visco-elastic nature and structure of skin is designed to maintain its integrity (Ryder et al, 1988). Tissue damage can occur due to altered soft tissue mechanics together with an increased magnitude and/or duration of shear, compressive or frictional forces. A simple rule of thumb to identify such tissue damage is to see if the skin can be depressed easily and it remains depressed. An appreciation of, and the ability to identify, the components that may contribute to such conditions is an important part of the screening process and intervention strategies.
Xeroderma
Xeroderma (dryness of the skin) caused by a decrease in moisture and lipid content within the stratum corneum is a common finding in people with diabetic neuropathy and is considered to be concomitant with the normal skin ageing processes (Pigatto et al, 1996; Nicander, 1988). Loss of moisture from the skin can result in functional deficits within both the dermis and epidermis and a reduction in its visco-elastic and anti-frictional qualities. Such functional losses can lead to callus formation (which can lead to ulceration) and skin fissures which can expose the dermis to potential pathogenic invasion, tissue loss and ulceration (Boulton et al, 1999). The frequent and regular use of bland emollients can help address this problem and additionally reinforces the importance of self-care and foot examination.
Fissures
Skin fissures are a common clinical finding occurring most frequently on the heel borders. They can deteriorate rapidly to ulcers and frequently become necrotic in patients with neuroischaemia (NICE, 2004). Deteriorated fissures must be dealt with swiftly and appropriately by referring the individual to a podiatrist for careful callus debridement at the fissure margins, which will allow the edges to knit together. Patients must be encouraged to apply bland emollients at least once daily to allow rehydration of the affected skin, preferably using a urea- or glycerine-based cream (Miettinen et al, 1999; Loden, 1996). The regular use of such emollients has been shown to reduce and replenish moisture loss, thus increasing suppleness (Loden, 1996).
Hard skin/callus
Plantar callus (Figure 1) is a common characteristic of the diabetic neuropathic foot. Such calluses are typically located over the toe apices and metatarsal heads and are generally thick and relatively soft (Bevans and Bowker, 1999). In contrast, calluses in the neuroischaemic or ischaemic foot are typically thin, dry, glassy and very hard. These may also be found over main weight-bearing areas but are particularly evident on the borders of the feet (Bevans and Bowker, 1999).
The presence of a bloodstained callus is highly predictive of ulceration being present below the callus. Upon removal of the callus an ulcer can be found in up to 80 % of cases (Rosen et al, 1985; Harkless and Dennis, 1987). Bloodstained calluses look like raspberry or blackcurrant jam under the skin. Individuals presenting with such calluses should be referred to a podiatrist for urgent debridement and pressure relief.
Fungal infections
Fungal skin and nail infections (Figure 2) in the foot can cause considerable discomfort and distress and have a reported prevalence of between 3 % and 8 % in the UK (Roberts, 1992; English et al, 1967; Howell et al, 1988); however, the precise prevalence in the population with diabetes is not known (see pages 194–200 for a study on fungal foot infections in diabetes). The common skin manifestations include interdigital fissuring, flaking, vesicular eruptions, inflammation and intense puritus while in onychomycosis (fungal infection of nails) the nails may be thickened, discoloured, friable and deformed.
Fungal infections of the skin are not a primary cause of foot ulceration; however, because they erode the epidermis there is an increased risk for subsequent bacterial infections. The most common site for such epidermal erosion to occur is between the toes, where bacterial infection develops quickly and may lead to deep ulceration and cellulitus (Yosipovitch et al, 1998; Semel and Goldin, 1996). It is therefore important to examine between patients’ toes during foot screening. Fungal infections of the skin can be treated quite easily by the topical application of anti-fungal creams (Bolognia and Braverman, 1998). Nail infections, however, are more difficult to treat and generally require systemic anti-fungal therapies sometimes in conjunction with topical anti-fungal nail lacquers.
Diabetic dermopathy
Diabetic dermopathy is the most common cutaneous finding in people with diabetes – appearing in 30 % of people with diabetes (Huntley, 1982). Diabetic dermopathy often results in pigmental patches/lesions which are often bilateral, multiple and hyper-pigmented macules on the pre-tibial areas (Sueki and Fujisawa, 1986). These patches are also known as shin spots. Lesions may also appear on the forearm, the side of the foot and the anterior surface of the lower thigh. The lesions are round or oval, reddish-brown, scaly papules and plaques, ranging in size from 0.5–1 cm. The incidence of diabetic dermopathy has been shown to correlate with an increase in glycosylated haemoglobin and duration of diabetes (Shemer et al, 1998).
Due to the frequent location of the lesions over bony prominences, diabetic dermopathy may simply be a magnified response to trauma such as heat, cold, or blunt objects in people with diabetes. There may be a link between diabetic dermopathy and other complications of diabetes such as microangiopathy and neuropathy (Jelinek, 1994). Thus, patients with diabetic dermopathy should be further evaluated for detection of other concomitant complications. To date, there is no effective treatment for diabetic dermopathy.
Bullosis diabeticorum
Bullosis diabeticorum, although rare, is a distinct complication of diabetes (Bolognia and Braverman, 1998; see Figure 3). The bullae (blisters) appear spontaneously from normal skin, usually on the feet and toes, but occasionally on the hands and fingers. They are usually 0.5 to several centimetres in size, and contain a clear, sterile, viscous fluid.
The bullae could be intra-epidermal or sub-epidermal. Intra-epidermal bullae are clear, sterile, non-haemorrhagic blisters that generally heal on their own within 2–5 weeks without scarring or atrophy. This type of bullae is more common in men with long-standing diabetes, peripheral neuropathy and good circulation to the involved extremity (Bolognia and Braverman, 1998).
Sub-epidermal bullae are the least common of the two types of bullosis diabeticorum (Huntley, 1982). These blisters are similar to the intra-epidermal blisters except they are occasionally haemorrhagic and may heal with scarring and atrophy. Bullosis diabeticorum is usually self-limiting; however, bacterial infections can result in deep infections (Huntley, 1982).
Local care to avoid blister eruption and appropriate antibiotic treatment for secondary infection is recommended.
Infection
Foot infections are common in patients with diabetes and sometimes are the presenting feature of undiagnosed type 2 diabetes. It is important to note that the cardinal signs of infection – redness, heat, swelling and pus – may be reduced and therefore are not as obvious as in those without diabetes. Unexplained raised blood glucose levels, sudden localised pain or discomfort in neuropathic ulcers can signify the onset of infection well before the presence of any clinical signs. Foot infections are serious and can progress very quickly, which, if not managed correctly and aggressively, can lead to lower-limb amputations. Any infection that does not show signs of responding to antibiotic therapy after 3 days should be treated very seriously and referred to a specialist foot clinic. Additionally, any patient with spreading cellulitis requires an immediate hospital referral as it is a limb-threatening condition (NICE, 2004).
Joints
Limited joint mobility
Collagen abnormalities – principally, thickening and increased cross-linking – are common in people with diabetes and are due to advanced glycation end-products (Goodfield and Millard, 1988). This is clinically evident by the skin having a tight and waxy appearance.
Testing for reduced joint mobility in the foot is important, but not very easy and requires a good understanding of foot anatomy and skill. Additionally, it can also be very time consuming in the hands of a novice. Reduced joint mobility in those with diabetic neuropathy has been shown to significantly increase foot pressures and the risk of ulceration (Fernando et al, 1991). As previously mentioned this is further increased when plantar callus is present.
Since limited joint mobility in the hands/wrists is sometimes indicative of joint problems in the feet and ankles (Ismail et al, 1996; Brik et al, 1991), perhaps the quickest way of screening for limited joint mobility is to ask the patient to perform the ‘prayer sign’. This is undertaken by asking patients to put the palms of their hands together with the fingers extended as if praying. If the palms do not meet the sign is said to be positive indicating limited joint mobility (Rosenbloom et al, 1981). Obviously this test is of little use if there are hand or finger deformities present such as Dupuytren’s contracture.
Perhaps two of the most important joints in the foot to examine are the ankle and big toe joint (first metatarso-phalangeal joint). This is because reduced ankle and first metatarso-phalangeal joint dorsiflexion increases the force and duration of loading at the forefoot and the plantar surface of the big toe respectively, which may increase ulceration risk in these areas.
Testing for reduced ankle joint dorsiflexion
Ankle joint equinus is deemed to be present when there is less than 10 º dorsiflexion from its neutral position.
With the lower leg fully extended, place your hand under the plantar surface of the foot so that the hand forms a rigid platform upon which the foot can rest. Gently push the foot upwards until it is at 90 º to the lower leg, not allowing it to roll in or out as you do so. From this position gently push a little harder to obtain the maximum amount of dorsiflexion as possible. There should be at least 10 º further dorsiflexion from this starting point; if this is not evident ankle equinus is likely to be present. This is a factor of high risk for ulcers in the presence of neuropathy (Fernando et al, 1991). Stretching exercises may be of some benefit.
First toe dorsiflexion
Gently grasp the base of the first toe between your thumb and forefinger with one hand and with the other do likewise just before the first metatarsal head. With the toe and first metatarsal head level, gently but firmly move the toe upwards. You should expect the toe to move up between 60 º–90 º from the starting point. If this cannot be achieved, the joint has a reduced range of movement. Once again this increases the risk of ulceration under the first toe especially in the presence of peripheral neuropathy (Fernando et al, 1991).
Excessive joint mobility
Although limited joint mobility is known to be associated with neuropathic ulceration, excessive joint mobility, especially at the mid-tarsal joint, can significantly alter forefoot time–pressure integrals and shear forces, thereby increasing the potential for callus formation and ulceration. The simplest way to examine for this is to observe what happens to the feet when the patient stands. If feet roll-in excessively or the medial arches flatten markedly it is most likely that the joints are very mobile.
In the author’s opinion this is a useful test to perform in order to determine the presence of motor neuropathy when high arches and clawed toes are observed at rest. If the toes remain clawed and the arches high upon standing, motor neuropathy can be strongly suspected; however, if the arches flatten motor neuropathy is unlikely even if the toes remain clawed.
Charcot neuroarthropathy
Although Charcot neuroarthropathy is not common it is important that it is identified early to help prevent devastating foot deformity and potential ulceration or amputation (Saunders and Frykberg, 1991). Typically, an active Charcot neuroarthropathy would present as a foot and leg that is intensely pink, warm and swollen with deformity that has occurred fairly suddenly (Jeffcoate et al, 2000; Jude and Boulton, 2001). Always compare the suspected foot and leg with the contra-lateral limb. Additionally, patients may describe pain which, if present, is generally a dull to intense aching and may be experienced in the foot or calf. Pulses will be readily palpable and abnormally high ankle systolic pressures are common due to calcification. Dilated dorsal foot veins are also often present. Charcot neuroarthropathy only occurs in patients with marked peripheral neuropathy and not in those with ischaemia.
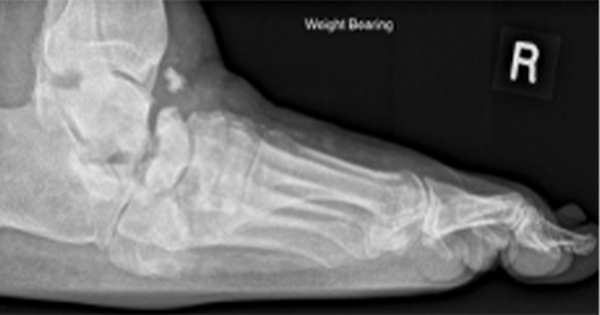
Foot deformity
Foot deformities (Figure 4) commonly occur as a result of altered foot mechanics, poorly fitting footwear, neuropathy and surgery. The most common deformities are clawed or hammered toes and hallux valgus (bunions). These are frequent causative factors for abrasions, callus, blisters and ulcers, due to shoe rubs both in patients with neuropathy and those with neuro-ischaemia. Thus footwear examination and advice to the patient is essential. It is important to remember that the shoes worn to clinics may not be worn most of the time – people commonly ‘dress to impress’.
Conclusion
Any advice that is given needs to be relevant and structured to each individual along with being realistically achievable. In conclusion, this article has tried to give an overview of some aspects of foot screening that involve observational skills rather than the use of diagnostic tools. Risk assessment and categorisation must involve combining a thorough clinical examination with diagnostic tests, balanced by good history taking. Recognising subtle skin or joint changes, which on their own may appear trivial but are major when combined with neuropathy or ischaemia, is very important. Combining such skills and expertise in diabetic foot screening is the secret to lesion prevention and good quality foot care. When examining feet, the opportunity should always be taken to educate the patient in order to reinforce the importance of self-care and identifying unresolved motivational barriers. Finally, it is hoped that this article, combined with the others in this series (Baker et al, 2005a; 2005b), should provide a ‘user’s guide to diabetes foot screening’.