What are incretin therapies and how do they work?
Incretin therapies mimic or enhance the action of gut hormones – primarily glucagon-like peptide-1 (GLP-1) and gastric inhibitory polypeptide (GIP) – to increase glucose-dependent insulin secretion, suppress glucagon release, slow gastric emptying and reduce appetite, leading to improved glycaemic control and weight loss.1
Semaglutide (Wegovy®) – GLP-1 receptor agonist1,2
Semaglutide is a long-acting GLP-1 receptor agonist (97% homology to native human GLP-1) that produces weight loss through multiple mechanisms.
- Appetite suppression via central nervous system GLP-1 receptors in appetite-regulating regions.
- Delayed gastric emptying, leading to increased satiety and reduced caloric intake.
- Glucose-dependent insulin secretion and suppression of inappropriately elevated glucagon.
- Improved glycaemic control in people with type 2 diabetes.
Tirzepatide (Mounjaro®) – dual GIP/GLP-1 receptor agonist)3,4
Tirzepatide is a first-in-class dual GIP and GLP-1 receptor agonist that produces superior weight loss compared with selective GLP-1 receptor agonists, through:
- Enhanced appetite suppression via dual receptor activation.
- GIP receptor activation: increases energy expenditure, improves insulin sensitivity and may reduce food intake.
- GLP-1 receptor activation: appetite suppression, delayed gastric emptying and improved glucose homeostasis.
- Complementary effects of GIP and GLP-1 pathways result in greater weight reduction than GLP-1 agonism.
Licensed indications2,3
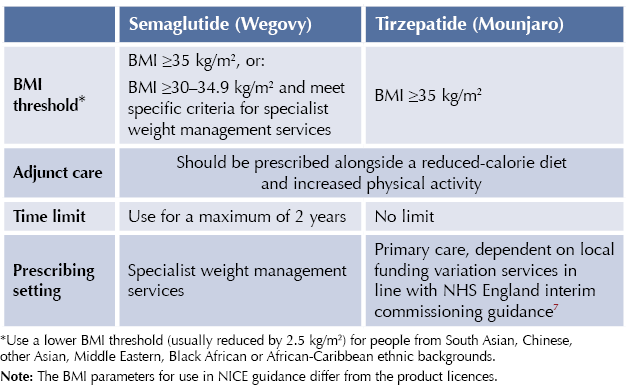
Subcutaneous semaglutide (Wegovy) and tirzepatide (Mounjaro) are indicated as adjuncts to a reduced-calorie diet and increased physical activity for weight management, including weight loss and weight maintenance, in adults with an initial BMI of:
- ≥30 kg/m2 (obesity), or
- ≥27 kg/m2 to <30 kg/m2 (overweight) in the presence of at least one weight-related comorbidity.
Review incretin therapy for weight management at 6 months and continue only if the individual demonstrates meaningful benefit with an acceptable risk–benefit profile.
Note that the oral formulation of semaglutide (Rybelsus®) is not licensed for weight management.
Position in NICE guidance
Medicines for weight management are recommended after dietary, exercise and behavioural approaches have been started and evaluated. All medicines for weight management should be used alongside a reduced-calorie diet and increased physical activity.
The NICE technology appraisals TA875 and TA1026 recommend tirzepatide and semaglutide as options for managing overweight and obesity in adults, only if all of the following conditions are met:5,6
Cautions and adverse events2,3
Gastrointestinal effects
● Most common adverse effects are gastrointestinal (nausea, vomiting, diarrhoea, constipation):
- Usually mild to moderate and transient.
- Occur more frequently during dose escalation.
● Advise patients to stay well hydrated.
● Consider slower titration or temporary dose reduction if symptoms persist.
Acute pancreatitis1,8
● Rare but serious adverse reaction observed with GLP-1 receptor agonists.
- Discontinue immediately if pancreatitis is suspected.
- Do not restart if pancreatitis is confirmed.
- Consider alternative aetiologies in people with persistent severe abdominal pain.
Hypoglycaemia
● Risk is low when used as monotherapy for obesity.
● Increased risk when used concomitantly with insulin or sulfonylureas in people with type 2 diabetes.
- Consider reducing dose of concomitant insulin or sulfonylurea.
- Educate on signs and symptoms of hypoglycaemia.
Diabetic retinopathy9
● Rapid improvement in glycaemic control may be associated with worsening of diabetic retinopathy.
- Monitor patients with pre-existing diabetic retinopathy.
- Use with caution in patients with history of diabetic retinopathy.
- See At a glance factsheet: GLP-1 receptor agonists and diabetic retinopathy for more information.10
Acute gallbladder disease
● Increased risk of cholelithiasis and cholecystitis observed.
- If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated.
Pregnancy and fertility
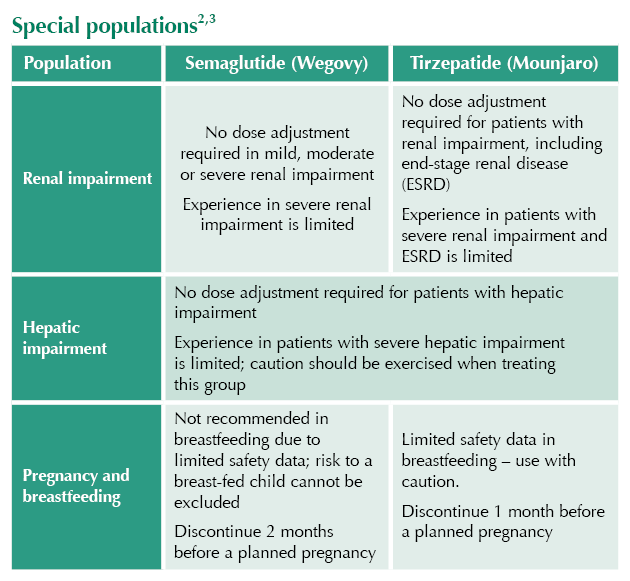
● Discontinue at least 2 months before planned pregnancy (semaglutide) or 4 weeks (tirzepatide), due to long half-lives.1,2
● Weight loss may increase fertility in anovulatory individuals.
● Advise females of childbearing potential to use effective contraception.
Thyroid cancer (in mice)
● Non-lethal thyroid C-cell tumours observed in rodents are a class effect; significance in humans is unknown.
Drug interactions and special considerations
Effect on gastric emptying and oral drug absorption2,3
● Both medications delay gastric emptying, which may affect absorption of concomitantly administered oral medications.
● Review all concurrent medications when initiating incretin therapies.
● For medications with time-dependent absorption, consider administering ≥1 hour before incretin injection.
● Monitor clinical response and therapeutic drug levels where applicable.
● Educate patients to report any changes in medication effectiveness.
Oral contraceptives and HRT11
● Delayed gastric emptying may affect absorption, particularly during initiation and dose escalation.
● Advise patients using oral contraceptives to switch to non-oral alternatives, or add a barrier method of contraception for 4 weeks after initiation and 4 weeks after each dose escalation.
● Similar recommendations are made to ensure the absorption and bioavailability of oral hormone replacement therapy.
- See Need to know for more information.12
Levothyroxine
● Monitor thyroid function tests when initiating or discontinuing incretin therapies in patients on levothyroxine.
● Changes in gastric emptying may affect levothyroxine absorption.
● Advise taking levothyroxine consistently relative to timing of incretin dose.
Narrow-therapeutic-index medications
● Exercise caution with medications that have a narrow therapeutic index:
● Digoxin: Monitor levels and clinical response.
● Immunosuppressants (e.g. tacrolimus, ciclosporin): Monitor levels closely.
● Antiepileptics: Monitor seizure control and drug levels where applicable.
● Warfarin: Monitor INR more frequently when initiating, titrating, or discontinuing incretin therapies in patients on warfarin.
Treatment discontinuation criteria
Consider discontinuation if:
● Weight loss is <5% of initial body weight at 6 months (per NICE guidance).3,4
● Intolerable adverse effects despite dose adjustment.
● Development of contraindication (e.g. pregnancy, pancreatitis).
● Patient request or struggling to adhere to regimen.
● Target weight achieved and maintained (individualised decision).
Dosing regimens

Missed doses
Semaglutide: If dose is missed and the next scheduled dose is >2 days away, administer as soon as possible; otherwise skip and resume normal schedule.
Tirzepatide: If a dose is missed, administer within 4 days; otherwise, skip and resume normal schedule.
Special populations2,3
Storage requirements
Before first use: Store in refrigerator (2–8°C). Do not freeze. Protect from light.
After first use: May be stored at room temperature (up to 30°C) for up to 30 days (tirzepatide) or 6 weeks (semaglutide). Keep pen cap on when not in use.
Available forms
Tirzepatide (Mounjaro): Pre-filled single-dose pens available in 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg.
Semaglutide (Wegovy): Pre-filled single-dose pens available in 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, and 2.4 mg.
Points to cover when counselling
□ Explain mechanism of action: appetite suppression and delayed gastric emptying.
□ Emphasise that medication is adjunctive to diet and exercise, not a standalone solution.
□ Discuss common gastrointestinal side effects and strategies to minimise (eat slowly, smaller portions).
□ Advise reporting persistent abdominal pain (pancreatitis warning).
□ Ensure understanding of injection technique and site rotation.
□ Discuss importance of contraception in females of childbearing potential.
□ Advise discontinuation 2 months (semaglutide) or 4 weeks (tirzepatide) before planned pregnancy.
□ Provide written information on thyroid cancer warning signs (neck lump, hoarseness, dysphagia).
□ Educate on potential drug interactions affecting oral medications.
□ Discuss realistic weight loss expectations (typically 10–20% body weight loss).
□ Provide information about monitoring requirements and follow-up schedule.





Findings should motivate us to encourage statin initiation and persistence for primary prevention in more people with type 2 diabetes.
26 Feb 2026