He was started on insulin therapy with multiple daily injections and received a Dexcom G6™ (Dexcom Inc, San Diego, CA, USA) for real-time continuous glucose monitoring (rtCGM). The insulin regimen chosen was 1.5 units of long-acting insulin analogue (Levemir) with breakfast and 2.5 units at bedtime; and meal-time fast-acting insulin aspart (Fiasp). His parents were given carbohydrate counting education and he was started on insulin aspart injection with ratios of 1:15 for breakfast, 1:16 for lunch and 1:21 for dinner. A month after starting insulin therapy, his HbA1c was 75 mmol/mol (11.7%).
There is a higher incidence of T1D in individuals with DS compared with the general population (Bergholdt et al, 2006, Rohrer et al, 2010). Furthermore, Bergholdt et al (2006) considered the prevalence of T1D in all children born in Denmark from 1981–2000 and found the prevalence of children with DS and T1D was 4.2 times higher than the prevalence of T1D in the background population. DS is also associated with a higher risk of developing other autoimmune conditions, such as coeliac disease and hypothyroidism (Lämmer et al, 2008; Aitken et al, 2013). In one study, the incidence of individuals with diabetes and DS who were diagnosed with thyroid disease and coeliac disease was 74% and 14%, respectively (Aitken et al, 2013). The child presented in this case study did not have a co-existing autoimmune condition at diagnosis, but there is an increased risk that he will develop one in the future.
Additional challenges
A diagnosis of T1D has a huge impact on the lives of both the child and their parents. It involves a lifetime of daily injections, monitoring glucose levels and accounting for every meal and snack eaten. For all families of a recently diagnosed child this is a challenging time (Lindström et al, 2011), but for the family of a child with DS, there are additional challenges for both them and the diabetes care teams (Pikora et al, 2014). The aim of diabetes care is to provide education and support for the child and family, and ultimately to empower the developing adolescent to become independent in their daily diabetes management. DS adds additional challenges to this process (McVilly et al, 2014; Pikora et al, 2014).
The characteristics associated with DS need to be considered by the diabetes team when implementing insulin therapy. Cognitive development varies between individuals with DS, particularly with regards to language, processing language and verbal working memory (Silverman, 2007; Couzens et al, 2012). Additionally, Couzens et al (2012) suggested that long-term medical conditions affecting daily lives were associated with a negative impact on cognitive ability and development of the child with DS. The child in this case report is still very young and it is unclear how his cognitive development will progress. He currently attends a mainstream primary school and both parents have university level education; these two factors have been shown to be supportive of cognitive growth (Couzens et al, 2012).
Learning disability (LD) associated with DS may also affect the communication of common T1D symptoms such as thirst, headaches, blurred vision and mood change. These may not be well communicated, making it more difficult to monitor and manage the condition (Taggart et al, 2013). LD does not preclude common emotions, such as feeling different and self-conscious when monitoring glucose levels in front of others (Dysch et al, 2012; McVilly et al, 2014). This could be the reason why LD has also been associated with sub-optimal glycaemic control (Taggart et al, 2013). This may not always be the case, however. In contrast, Rohrer et al (2010) found individuals with DS and T1D achieved better HbA1c and used less insulin than people with T1D without DS, despite their intellectual impairment. The authors speculated that this may be due to their simpler lifestyle and acceptance of routine. We should be mindful of these possibilities as the child develops.
Continuous glucose monitoring
The diabetes team opted to use continuous glucose monitoring (CGM) to overcome some of the issues of more challenging glycaemic control associated with LD. CGM is beneficial for children and young people (CYP) who have an impaired awareness of hypoglycaemia, or are unable to articulate symptoms of hypoglycaemia or hyperglycaemia, as was the situation with the child in this case study (Danne et al, 2018). The internationally agreed target range for glucose levels is 3.9–10 mmol/L (Battelino et al, 2019) and CGM has been shown to improve “time in range”, regardless of the type of insulin therapy used.
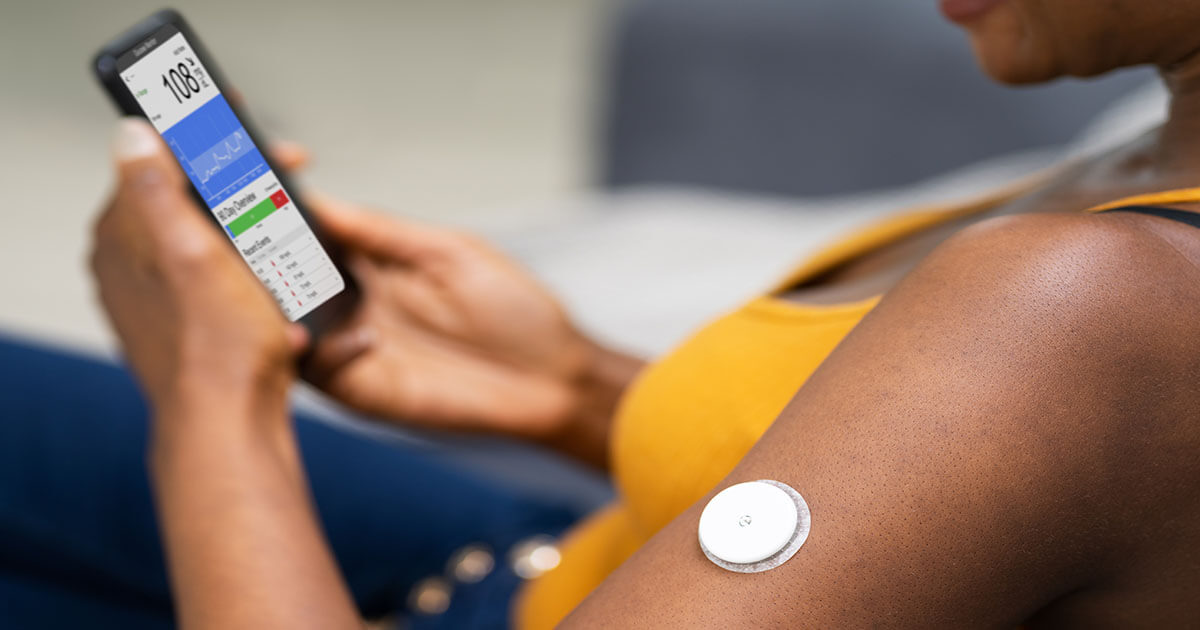
The Dexcom G6 was chosen because it offers rtCGM and has been shown to increase time in range from 47.4% to 57.0% in children, when combined with a structured education programme (Pemberton et al, 2020). NICE (2015) recommends rtCGM with a hypoglycaemia alarm system for children with an inability to recognise and communicate the symptoms of hypoglycaemia. Puhr et al (2019) found the switch to Dexcom G6 with hypoglycaemia alarms reduced the time in hypoglycaemia by 40% for those with a hypoglycaemia threshold setting of 3.9 mmol/L, and by 33.3% for those with a threshold setting of 4.4 mmol/L. Pemberton et al (2020) report that the Dexcom G6 is currently the only device available in the UK which meets the Food and Drug Administration requirements for use in the ages 2–70 years, has four hypoglycaemia prevention alarms and does not require frequent finger-prick calibrations.
In the current case it was thought that, based on his age and current cognitive development, this child might not recognise and be able to communicate symptoms associated with low blood glucose levels (Taggart et al, 2013). The Dexcom G6 offers a solution to this and enables his parents to monitor real-time data, as well as reducing the frequency of finger-prick blood glucose tests. As with all children with T1D, DS does not reduce the perceived impact on daily life, and children with DS may still have a needle phobia (Pikora et al, 2014). His parents have found the ability to monitor his glucose levels throughout the day when he is at school reassuring. In addition, rtCGM allows overnight monitoring for nocturnal hypoglycaemia.
Fast-acting insulin
This child has a regular meal pattern and routine regarding his meal choices. Following discussions with his parents regarding meal patterns, a view was taken that although he tended to eat from a limited range of meals, the actual portion size of his meal varied and was often difficult to predict until during, or even after, the meal. The team decided that the most appropriate bolus insulin would be Fiasp, which has been shown to start acting 5 minutes earlier than insulin aspart (Heise et al, 2017; Russell-Jones et al, 2017; Shiramoto et al, 2018). The speed of onset offered the parents more flexibility at mealtimes to accommodate the fluctuating appetite of a five-year-old.
Diabetes education
The aim of education for CYP with T1D is to empower children and adolescents to manage their own diabetes according to their chosen management plan (Phelan et al, 2018). The same is true of education for children with special educational needs and LD (Dysch et al, 2012; McVilly et al, 2014). DS is associated with delays in overall cognitive development and language development (Finestack and Abbeduto, 2010; Quinn et al, 2020). As a team, we will need to adapt our style of education to reflect this child’s cognitive learning. This will involve working with the family to ensure that all diabetes-related education is unambiguous and to establish their preferred style of communication.
Augmentative and alternative communication
As a team, we will need to consider different teaching strategies to help engage the child to participate in his own care as he gets older. One such strategy is augmentative and alternative communication (AAC). AAC is often used to support CYP with cognitive disabilities, including children with DS. Symbols are used to teach instructions and to communicate needs (Binger and Light, 2007; Finke et al, 2017; Quinn et al, 2020).
Multi-symbol messages and using symbols in different combinations can convey simple requests and tasks, such as “more milk” or “drink milk” (Binger and Light, 2007). AAC modelling (AAC-MOD) involves a combination of symbols while simultaneously modelling the spoken messages and is used to aid the child’s understanding of those messages (Quinn et al, 2020). AAC-MOD provides a method of communication to aid participation in daily routines.
Similarly, objects could be used as an alternative to symbols. Diabetes-related objects of reference could include an empty insulin pen with a symbol prior to receiving an injection, so the child understands what is about to happen. Symbols could then be used to create a schedule to help this child understand what he needs to do throughout the day to manage his diabetes. These could be combined with a task schedule board. When a task is completed, it is removed from the schedule board. This can help remind a person which tasks have been completed and which are still to be done. At the time of writing, AAC-MOD had not yet been implemented in practice.
School
Children spend a significant proportion of their day at school, and maintaining glucose levels as near normal as possible is important in order to optimise their learning ability (Bratina et al, 2018). Schools in England should support children with medical conditions, so that they have full access to education (Department of Education, 2015). In pupils with medical conditions who have special education needs, this provision should be delivered in a coordinated way (Department of Education, Department of Health, 2015). Education, Health and Care Plans (EHCPs) are used to identify additional needs and are designed to increase collaboration between education, health and care teams (Boesley and Crane, 2018).
This child already had a comprehensive EHCP due to his pre-existing healthcare needs and has one-to-one support from a teaching assistant (TA) throughout the school day. This has enabled the school to accommodate his additional care needs as a result of diabetes. For example, his parents requested his insulin injections to be given in his classroom rather than in the school office, as had been the case with previous pupils with T1D. His mother describes him as stubborn and knew he could potentially refuse to go to the office. To minimise the negative associations with his diabetes, it was agreed his injections could be administered in the lobby area of his classroom. Due to the fact that this child had one-to-one support already in place through his existing EHCP, the school felt able to accommodate this and the additional medical care required as a result of his new diagnosis of T1D.
This child’s response to insulin injections varies and he will sometimes say “no, don’t hurt me” to his TA. To address concerns that his classmates might be distressed by this, parental permission was obtained for the Year 1 children to watch the BBC Get Well Soon episode “Learning more about diabetes”. This enabled the children to ask questions and was well received by the parents. His mother reports that he is comfortable engaging in his care in the classroom and his classmates appear happy to chat openly about it.
This child’s current primary school is working with his parents and healthcare team to tailor his support to meet his specific needs, and it has the resources to do so. As he progresses through secondary education, the healthcare team need to be cognizant that this may not always be the case. They will need to continue to work collaboratively with future schools to ensure this tailored support can continue where practicable.
Future challenges
As a team we will need to develop our service and practice over the coming years to ensure this child has the support he needs to manage his diabetes. In the meantime, there is a paucity of studies about DS and the management of T1D, and McVilly et al (2014) suggest further research is needed to determine the type of support techniques and organisational support that are required to maximise active involvement by individuals with LD in the self-management of their diabetes. Whichever strategies are chosen, it will require ongoing collaboration, clear communication and coordinated planning between us, the healthcare team, his family, school staff and, at a later stage, the individual himself, to ensure he gains the support he needs and is empowered to manage his diabetes care as independently as possible.
Acknowledgement
The author would like to acknowledge Birmingham City University’s Children & Young Persons Diabetes Care Module.





NHSEI National Clinical Lead for Diabetes in Children and Young People, Fulya Mehta, outlines the areas of focus for improving paediatric diabetes care.
16 Nov 2022