The discovery and increased understanding of biofilm over the past decade has changed approaches to wound care. Biofilms are communities of bacteria encased in a matrix of polysaccharides, protein and DNA, which provide high levels of resistance to antimicrobials while mitigating the effectiveness of elements of the host’s natural immune system, such as macrophages. There is evidence that >80% of all biopsies performed on chronic wounds have a biofilm, whereas only 6% of acute wounds do, suggesting a strong correlation between biofilms and delayed healing (Malone et al, 2017). It is understood that biofilms impair healing by stimulating chronic inflammation, leading to elevated levels of proteases and reactive oxygen species (ROS) that degrade proteins essential for healing.
William explained: “We know that biofilms are hard to penetrate; you have to soak them in stabilised hypochlorous acid for quite a while, between 10–30 minutes. So really, the key here is sharp surgical debridement.” A lack of aggressive intervention to remove biofilms can be regarded as one of the key barriers to initiating healing in chronic wounds.
“We need to be more aggressive with debridement. What that means in practice is that if you notice in the community setting that a wound is not progressing to closure, bring the patient in and get the non-viable tissue surgically removed,” urged William. In the US, telehealth services have been set up to support community nurses with a degree of success, yet some patients remain reluctant to receive hospital-based treatment.
While debridement is a critical first step in biofilm-based wound care, biofilms can reform quickly (~3–7 days) so adequate wound bed debridement needs to be repeated weekly and be combined with targeted treatment. William recommended a step-down-step-up (SD-SU) approach (Figure 1). SD-SU therapy is based on starting with the therapies that most effectively reduce biofilms, inflammation and proteases (step-down), then shifting to advanced therapies with regenerative properties (step-up) that enhance repair of the wound bed, including dHACM (growth factors, protease inhibitors, intact collagen).
There are some crucial considerations to the success of this approach, such as when to initiate negative pressure wound therapy (NPWT). “If you’re doing deep debridement and getting near to bone or other deep structures and you don’t utilise NPWT right away, you can possibly lose the remaining soft tissue, which ultimately places the patient at greater risk of an undesirable outcome,” William stated.
The impact of delayed healing
Often, autolytic debridement is the approach most favoured in the community setting, although it has come under increasing scrutiny as a slow and often ineffective approach.
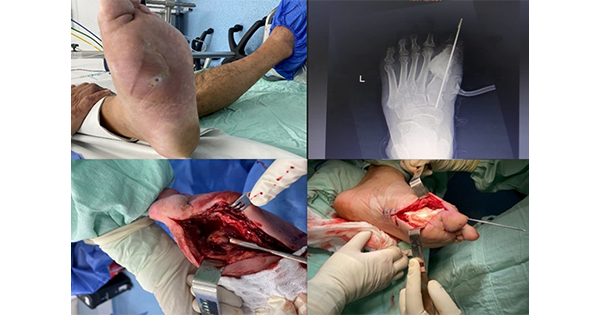
“We really need to reconsider this approach in favour of more surgical debridement,” said William, giving the example of a 50-year-old male patient (Figure 2) who had been failing on autolytic debridement. The patient had experienced a biking accident resulting in a leg ulcer worsening over the course of 1 month.
He added: “Where there are sclerosed or epibole of margins around a wound, you have to open those up; sometimes you need to make a wound slightly larger to get it to heal. You want the immature epidermal cells that originate in the basale stratum layer of the skin to migrate into the wound bed, to help close the wound.” The only way to encourage this is to open up the wound bed edges.
Delayed healing puts patients at serious risk of developing complications leading to increased morbidity, including amputations. “Thirty days is a defining chronic wound metric and a danger zone for patients,” William remarked. Evidence demonstrates wounds that are present for >30 days are 4.7 times more likely to become infected (Lavery et al, 2006). Patients with infected wounds are 56 times more likely to be admitted to hospital, putting them at risk of potential hospital-associated complications (Lavery et al, 2006). Additionally, infected chronic diabetic foot ulcer patients are 155 times more likely to have an amputation (Lavery et al, 2006), proving why it is so critical to screen patients for infection and biofilm early, and to treat early “not with topical antibiotics, but with antiseptics, such as iodine, stabilised hypochlorous acid solutions or silver-based products, which do necessarily drive the emergence of resistant bacteria”. There are also significant cost implications associated with preventable hospitalisations and complications.
Elsewhere, evidence points to degree of closure over time as an indicator of outcomes. A multicentre, prospective randomised controlled trial (RCT) of 276 patients with diabetic foot ulcers (DFUs) found that if the wound size is not decreased by 53% at 4 weeks, the likelihood of healing at 12 weeks is only 9% (P<0.001) (Sheehan et al, 2003). “This is a great indicator to use with community nurses and a robust measure for escalating care. It could represent a big shift for improving outcomes,” William suggested.
Introduction to dHACM allografts
dHACM allografts are “changing healing,” said William. The technology behind dHACM allografts, which involves harvesting human placental and amniotic tissue, is not new. Natural human amniotic membrane has been used as a wound covering for more than 100 years (John, 2003). Human amnion is known to be immune-privileged, reduce inflammation, pain and scarring, provide a matrix for cell migration, as well as a natural biological barrier (Koob et al, 2013). First described in the literature in 1910, dHACMs were widely used in the treatment of burns up until 1960s. Thereafter, outbreaks of HIV and Hepatitis C rendered dHACMs ‘unsafe’ due to a lack of an effective screening method and potential for disease transmission. However, in recent years, a system has been identified to gently process, sterilise and dry placental tissue obtained from screened and tested pregnant women scheduled to undergo Caesarean delivery (Zelen et al, 2013) and, thus, dHACM is regaining popularity.
Today, donors are screened prior to and after delivery for infectious diseases to ensure tissue safety. The Purion® process, a validated process with proven bacterial/spore reduction capabilities (ranging from 1.4–5.6 Logs) is then used to remove a degree of bacteria from the harvested tissue. This Purion process allows for biological and structural integrity as the proteins in the tissue are preserved. Finally, the tissue undergoes another sterilisation step involving terminal radiation, thus further reducing the risk of transmitting an undetected emerging viral pathogen, such as Zika virus. Double sterile barrier product packaging ensures sterility is maintained post-distribution. Lastly, the product can be stored at an ambient temperature for approximately 5 years, making it economically viable by reducing waste and paperwork.
EpiFix: a solution for when standard of care fails?
The therapeutic potential of human amnion/chorion tissue grafts in wound healing is well established (John, 2003). EpiFix is derived from amnion/chorion tissue and has been shown to stimulate healing in hard-to-heal wounds, with potential to positively affect four distinct and pivotal physiological processes intimately involved in wound healing: cell proliferation, inflammation, metalloproteinase activity and recruitment of progenitor cells (Koob et al, 2013).
In the inflammatory phase of healing, matrix metalloproteinases (MMP) levels increase, but should peak at day 3 before returning to baseline by day 10. Chronic inflammation can lead to persistently elevated levels of MMPs, which are known to degrade both the excellular matrix and the messaging between cells that are required to progress through the phases of healing. EpiFix contains essential regulatory proteins that can suppress MMPs and promote healing in the wound bed (Koob et al, 2013).
A series of RCTs demonstrate the healing potential of EpiFix (a summary of evidence is provided in Table 1). In one study, when compared with standard of care (SOC) alone, EpiFix was shown to heal 92% of DFUs at 6 weeks, versus just 8% for the control (P<0.001). Futhermore, of the 92% of wounds that healed, 50% did so within 7 days of application of EpiFix (Zelen et al, 2013) (Figure 3).
Another trial comparing EpiFix with SOC and an alternative living product (Apligraf®, Novartis®) showed superiority for complete healing in DFUs at weeks 4, 6 and 12 (Zelen et al, 2015) (Figure 4).
A more recent RCT (Tettlebach et al, 2019) of 110 patients from 14 wound clinics confirmed the efficacy of EpiFix compared with SOC (sharp debridement, standard wound dressings with alginate dressings, absorbent non-adhesive hydropolymer secondary dressings and gauze alone). Time to complete wound closure was assessed over 12 weeks in patients with non-healing DFUs. In the intent-to-treat (ITT) group, 70% of patients who received weekly EpiFix plus SOC had complete healing by 12 weeks, compared with 50% in the SOC group (P=0.0338). Subjects that completed the study without a protocol deviation, the per-protocol (PP) group, demonstrated 81% of patients who received weekly EpiFix plus SOC had complete healing by 12 weeks, compared with 55% in the SOC group (P=0.0093). Furthermore, patients identified in the ITT cohort as having ‘inadequate debridement’ were 64% less likely to heal within 12 weeks, when controlling for covariates (P=0.022) (Tettlebach et al, 2019).
Conclusion
The use of sharp surgical debridement combined with the application of dHACM allograft (or known in the trade as EpiFix) over stalled wounds has been shown to significantly enhance healing rates in hard-to-heal wounds. Furthermore, inadequate debridement significantly impacts the likelihood of healing, suggesting patients could benefit from a more aggressive and proactive surgical approach.